Emerging From the Haze™: Pilot Feasibility Study Comparing Two Virtual Formats of a Cognitive Rehabilitation Intervention
Objectives: To gather feasibility and preliminary data comparing two virtual delivery methods for providing Emerging From the Haze™ (Haze) to cancer survivors compared to waitlist control (WLC).
Sample & Setting: Eligible participants (N = 93) reported cancer-related cognitive impairment following chemotherapy for stage I–III solid tumors, Hodgkin lymphoma, or non-Hodgkin lymphoma.
Methods & Variables: A three-arm randomized design was used to compare virtual live group presentation of Haze sessions, virtual prerecorded Haze group sessions, and WLC. Data were collected at baseline, week 10, and week 14.
Results: Feasibility was demonstrated. Significant cognitive function improvement at week 10 versus WLC was reported for the live group, and clinical improvement was reported for the prerecorded group. The prerecorded group reported significant improvement at week 14 versus WLC in physical activity, sleep, and health-related quality of life.
Implications for Nursing: Additional pilot and feasibility evidence for cognitive rehabilitation interventions was demonstrated. Prerecorded Haze delivery shows potential for clinical effectiveness and scalability. Future multisite research is warranted.
Jump to a section
The cognitive sequelae of cancer and cancer-related treatment for non–central nervous system malignancies, referred to as cancer-related cognitive impairment (CRCI), occur in as many as 75% of patients with cancer during treatment and continue for years post-treatment in about 35% of these patients (Ahles & Root, 2018; Ahles & Saykin, 2007; Henneghan et al., 2021, 2023; Janelsins et al., 2014; Koppelmans et al., 2014; Wefel et al., 2004). CRCI can be extremely distressing and may profoundly affect health-related quality of life (HRQOL), occupational achievement, social reintegration, and identity (Ahles et al., 2012; Boykoff et al., 2009; Crouch et al., 2022; Henderson et al., 2019; Myers, 2012; Von Ah et al., 2012). Research designed to mitigate the cognitive effects of cancer and cancer therapy is a key component of the Oncology Nursing Society Research Agenda’s priority area of palliative care and psychosocial oncology, particularly regarding the need to “determine the most effective interventions to improve patient and caregiver HRQOL” (Von Ah et al., 2019, p. 660)
Effective, evidence-based options for managing or treating CRCI that can be widely disseminated are extremely limited and urgently needed. Research to address this knowledge gap is crucial to promote HRQOL for cancer survivors. The efficacy of pharmacologic treatments has been inconclusive in this population, and these treatments are unlikely to address the wide-ranging and multifaceted concerns related to CRCI (Allen et al., 2018; Asher & Myers, 2015). Cognitive rehabilitation is an umbrella term that encompasses cognitive training, strategy training, and cognitive behavioral therapy (Dos Santos et al., 2020; Fernandes et al., 2019; Lange et al., 2019). Research indicates that cognitive rehabilitation and interventions targeting improvements in physical activity, sleep disturbance, mindfulness, and reduction of loneliness may improve cognitive function (Cacioppo & Hawkley, 2009; Chiesa et al., 2011; Jaremka et al., 2014; Johns et al., 2016; Lange et al., 2019; National Comprehensive Cancer Network, 2023; Von Ah & Crouch, 2020).
Emerging From the Haze™ (Haze) is a standardized, multidimensional cognitive rehabilitation intervention developed for cancer survivors experiencing CRCI. Haze consists of psychoeducational and experiential content and has been provided regularly within the cancer survivorship program at Cedars-Sinai Medical Center (CSMC) in Los Angeles, California, since 2010. Haze content is provided in a group setting by an interprofessional team with oncology rehabilitation, neuropsychology, and group facilitation expertise. Haze has shown promising results for improving patient-reported cognitive function in patients with a variety of cancer types who report CRCI following primary therapy for cancer (Asher et al., 2019; Liang et al., 2018; Myers et al., 2020, 2022). Haze initially was designed for only live, in-person delivery in six weekly 2.5-hour sessions. Retrospective analyses (N = 110) demonstrated significant improvements in perceived cognitive function (PCF) for participants (Asher et al., 2019). Positive results from previous waitlist-controlled pilot work demonstrated improvements in PCF for in-person groups of breast cancer survivors (N = 61) receiving Haze content live via videoconferencing (Myers et al., 2020). Subsequent positive results also were demonstrated from a single-arm investigation of virtual live delivery resulting the need for social distancing during the COVID-19 pandemic (N = 37) (Myers et al., 2022). Participants attended Haze in virtual groups instead of in-person groups. Based on participant feedback from the single-arm pilot, Haze was revised to provide the content in 10 shorter weekly sessions (60–90 minutes). However, virtual live delivery of Haze is restricted to the day of the week and time of day that the program is being delivered, which limits access. Development of a prerecorded format for Haze was of significant interest as a potential mechanism to increase scalability and facilitate eventual broad dissemination across settings and locations.
The purpose of this pilot study was to gather preliminary data comparing two telehealth virtual delivery methods to waitlist control (WLC) for the Haze standardized cognitive rehabilitation intervention for cancer survivors. The primary aim was to demonstrate the feasibility of a three-arm study design comparing the following groups: (a) virtual live Haze group sessions, (b) virtual prerecorded Haze group sessions, and (c) WLC. Secondary aims were to assess between-group differences for changes in PCF and to explore between-group differences for changes in health behaviors (physical activity, sleep, mindfulness), psychosocial outcomes (mood, loneliness), a determinant of behavior change (intention to change), and HRQOL.
Methods
Theoretical Framework
Two conceptual models guided this study. The Revised Conceptual Model of Chemotherapy-Related Changes in Cognitive Function based on the theory of unpleasant symptoms incorporates the impact of situational factors related to lifestyle and personal experiences, as well as the concurrent experiences of multiple symptoms, on changes in cognitive function in cancer survivors (Hess & Insel, 2007; Myers, 2009). This model depicts the potential relationships among the following study variables: physical activity (exercise), mood (anxiety and depression), loneliness (social support), PCF (self-reported), and the potential confounding variables of age, education, hormone status (e.g., menopause), and ongoing endocrine therapy. The study also was undergirded by the theory of planned behavior (TPB). The TPB indicates that the following four constructs are key determinants of behavioral change: (a) instrumental and affective attitudes toward the desired behavior, (b) perceived behavioral control, (c) subjective norms, and (d) intention to change the behavior (Ajzen, 1991, 2006; Ajzen & Fishbein, 1980). Intention to change has been shown to be the most proximal determinant of behavior change (Rhodes & Courneya, 2004) and was measured for the following three aspects of the cognitive rehabilitation intervention content: strategies to facilitate exercise, sleep quality, and mindfulness.
Study Design, Setting, and Sample
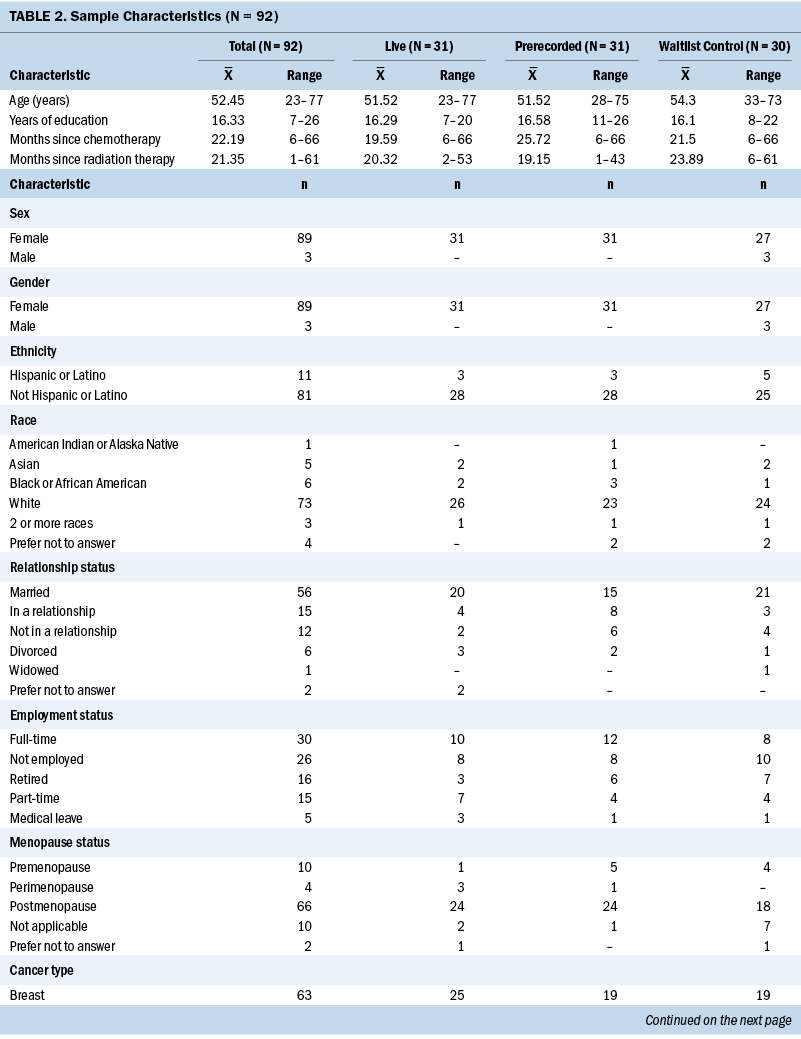
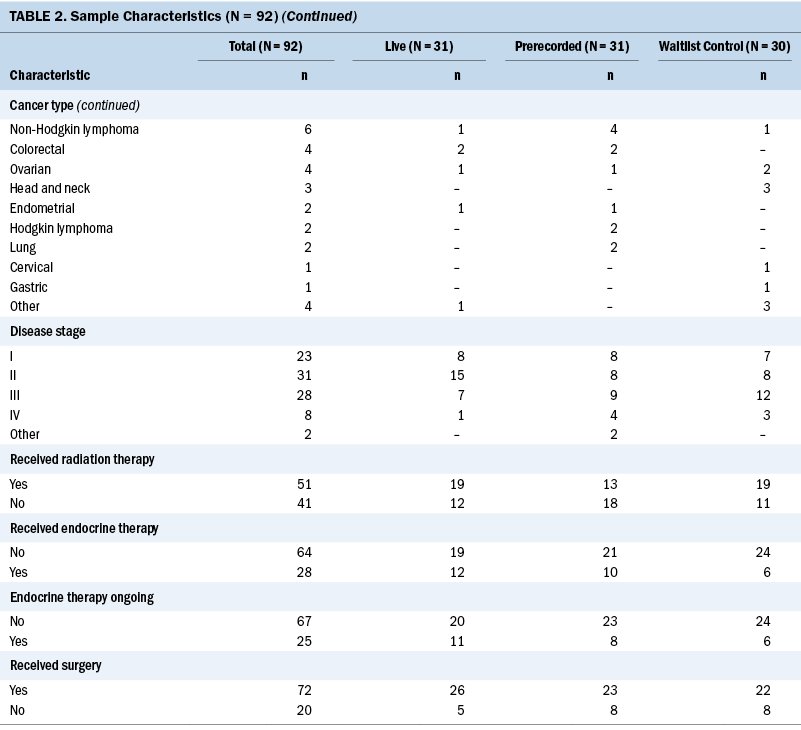
This prospective, randomized, waitlist-controlled, three-arm pilot study was conducted at the University of Kansas Cancer Center (KUCC) in Kansas City and CSMC. Eligible participants were adults (aged 18 years or older) diagnosed with stage I–III solid tumors, Hodgkin lymphoma, or non-Hodgkin lymphoma who were within six months to five years of completing chemotherapy (and radiation therapy if received) and who reported changes in cognitive function. These time frames were selected to be consistent with the researchers’ previous work (Myers et al., 2020, 2022) and to allow resolution of most acute treatment-related side effects, while still capturing shorter- and longer-term issues with cognitive function. Ongoing treatment with endocrine, anti-HER2, or other stable maintenance therapies (e.g., rituximab) were allowed. Patients were excluded for known history of other neurologic conditions involving impaired cognitive function (e.g., Parkinson disease, multiple sclerosis, Alzheimer disease or related dementia). Patients also were excluded for previous receipt of intrathecal chemotherapy or nonautologous marrow or stem cell transplantation (autologous transplantation was allowed). Feasibility was defined as successful recruitment (more than 95% of target sample), a greater-than-70% retention rate per group, 85% session attendance adherence, and positive participant satisfaction ratings. To achieve the primary aim for demonstration of feasibility, a planned sample size of 90 with 30 participants per group allowed for an attrition rate of as much as 20%. This sample size is consistent with recommended parameters for pilot studies (Browne, 1995; Julious, 2005; Teare et al., 2014). Twenty-four participants per group provided 80% power (95% confidence intervals estimated within 0.8 SDs) to detect a large between-group effect size (Cohen’s d = 0.83) using a two-sided, two-sample t test at the 0.05 level of significance.
Intervention
The intervention was designed and implemented by members of the study team. An overview of the Haze content is depicted in Table 1. The content is based on recommendations from the National Comprehensive Cancer Network (2023) for cancer survivorship and includes evidence-based information regarding behaviors related to brain health. Behaviors associated with the reduction of inflammatory cytokines and increase of markers of brain health like brain-derived neurotrophic factor are emphasized (Di Liegro et al., 2019; Lange et al., 2019). In particular, participants are encouraged to find sustainable ways to increase physical activity (target 150 minutes of moderate weekly exercise), decrease intake of processed foods and added sugars, increase intake of foods rich in omega-3 fatty acids, practice mindfulness strategies, and incorporate elements of sleep hygiene to reduce sleep disturbance and fatigue. Participants are led through a series of exercises and strategies to help increase concentration and focus, minimize distractions, and boost short-term memory. Content also is devoted to the management of negative thoughts that can exacerbate issues with cognitive function. All participants in the intervention group received paper and electronic copies of the program slides, handouts, and homework exercises.
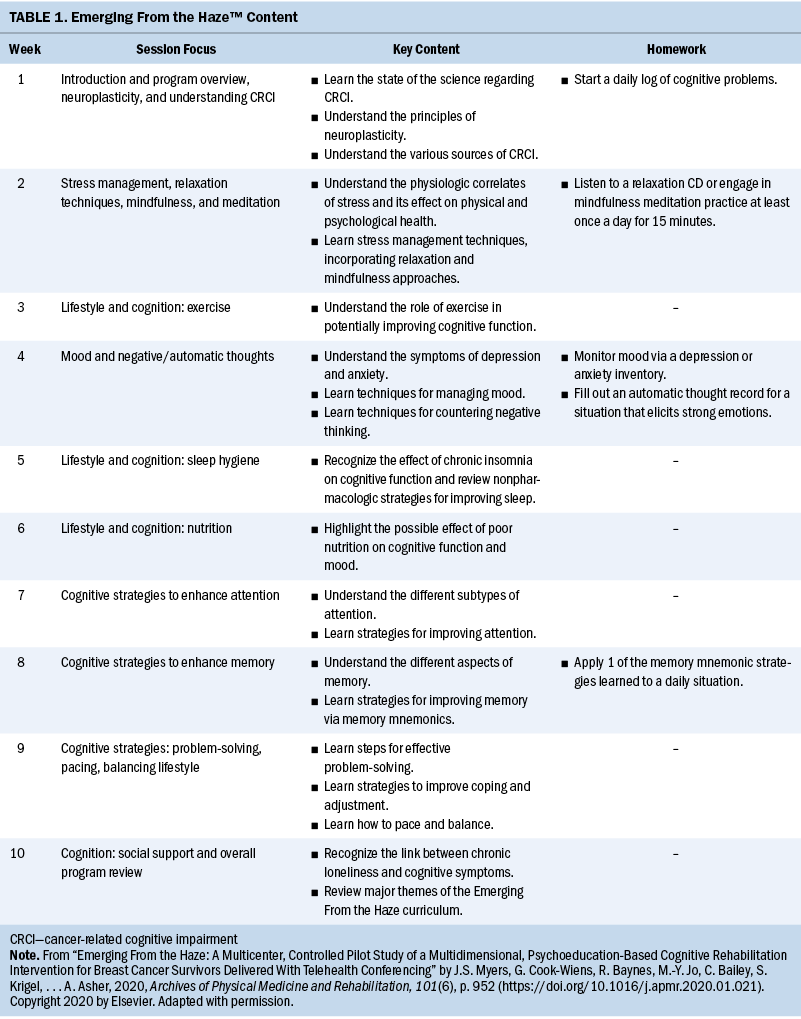
The virtual live and prerecorded groups were facilitated concurrently. Presenters of the virtual prerecorded program content included a physical medicine and rehabilitation physician (i.e., physiatrist) specializing in cancer rehabilitation and a neuropsychologist, thus ensuring content fidelity. Four cohorts of 10-week sessions were provided. Secure telehealth conferencing platforms were used for the live and prerecorded audio and video delivery of the intervention. The virtual live group webinar presentations (group 1) were broadcast in real time from CSMC. This same content was video recorded and stored on a secure shared drive provided by CSMC for this study. The prerecorded group sessions (group 2) were accessed from the shared drive and hosted in real time by the telemedicine and telehealth systems coordinator at the University of Kansas Medical Center, the umbrella academic association under which the KUCC operates. These prerecorded videos were synchronously watched by group 2 participants. Group 2 discussion was facilitated by one advanced practice oncology nurse to ensure intervention consistency. Participants in both virtual groups were able to ask questions in real time and take part in group discussions regarding the content. Participants in the WLC (group 3) were invited to attend their preferred delivery method after completing the study assessment time points for data collection.
To additionally ensure intervention fidelity, make any midstream study adjustments, and inform future study design, the study team met during and after each cohort to discuss participants’ satisfaction survey responses and troubleshoot any challenges related to technology, participants’ questions, or issues with data collection.
Recruitment
Following University of Kansas Medical Center Institutional Review Board approval, several recruitment strategies were employed. Individuals on the CSMC waitlist to attend the Haze program were approached regarding interest in study participation. In addition, patients reporting cognitive issues to the healthcare teams at CSMC and KUCC were provided information about the study. External referrals were received from other institutions (e.g., Swedish Cancer Institute, University of Michigan, Masonic Cancer Alliance membership). The Curated Cancer Clinical Outcomes Database was used to identify eligible patients receiving care at KUCC who had opted in or not opted out for being contacted about clinical trials.
Data Collection and Study Procedures
Informed consent was obtained prior to any data collection. In-person or virtual consent was allowed. Block randomization (blocks of six) was used via REDCap at the time of consent to ensure relatively equal group size for comparison (Harris et al., 2009, 2019). Data were collected at baseline (T1), immediately postintervention (week 10, T2), and week 14 (T3). Commensurate assessment time points were employed for WLC participants. Because this study was unblinded, participants were informed of their group assignment following informed consent and completion of the T1 study questionnaires to minimize any expectancy effect. Participants received a small payment incentive after completing all three sets of study questionnaires. Study questionnaires were administered electronically via REDCap. Cellular data plan–enabled, password-protected study tablets were loaned to participants who did not have internet access, videoconferencing-compatible devices, or reliable cellular service to ensure broad study access. Only deidentified data were extracted from REDCap for analyses. All data were stored on a secure, dedicated research drive at the University of Kansas Medical Center.
Instruments
Information from patient health records provided type and stage of cancer, treatment regimen(s), and pertinent medications. Study instruments required about 20 minutes to complete. The demographics questionnaire yielded data on age, sex, gender, race and ethnicity, years of education, menopause status, relationship status, and employment status. Other instruments were the Patient-Reported Outcomes Measurement Information System (PROMIS) Cognitive Function Short Form (CF), version 2.0; PROMIS Cognitive Function–Abilities Short Form (CF-A), version 2.0; Godin–Shephard Leisure-Time Physical Activity Questionnaire (GSLTPAQ); Pittsburgh Sleep Quality Index (PSQI); Mindful Attention Awareness Scale; PROMIS Emotional Distress–Anxiety, version 1.0; PROMIS Emotional Distress–Depression, version 1.0; University of California, Los Angeles (UCLA), Loneliness Scale, version 3.0; determinants of behavior change (intention) based on the TPB; PROMIS Global Health, version 1.2; and a satisfaction survey.
PROMIS CF and PROMIS CF-A: The PROMIS CF and CF-A were derived from the Functional Assessment of Cancer Therapy–Cognition item bank and designed to elicit self-reporting of cognitive function from cancer survivors. These instruments have been recommended for use in CRCI research by the Cancer Neuroscience Initiative Working Group (Henneghan et al., 2021, 2023). The PROMIS CF items are framed in a negative way (e.g., “My thinking has been slow”), and the PROMIS CF-A items are framed as positive statements (e.g., “My mind has been as sharp as usual”). The PROMIS CF is recommended (Henneghan et al., 2021) as the primary instrument of choice to harmonize data across studies and is the primary measure of PCF in this study. However, evidence has shown that use of the PROMIS CF-A as the secondary measure of PCF in this study may be of benefit because the two forms may measure two different constructs (Henneghan et al., 2021; Lai et al., 2014). Both instruments contain eight items. Items are ranked on a five-point Likert-type scale, with scores on the PROMIS CF ranging from 1 (very often) to 5 (never) and scores on the PROMIS CF-A ranging from 1 (not at all) to 5 (very much). Higher scores on each instrument reflect better cognitive function. Raw scores are rescaled into T scores with a mean of 50 and an SD of 10. Test–retest analyses from a single-arm pilot study indicated high reliability ratings for the PROMIS CF (Cronbach’s alpha = 0.946) and CF-A (Cronbach’s alpha = 0.952) (Myers et al., 2022).
GSLTPAQ: The GSLTPAQ, also called the Godin Leisure-Time Exercise Questionnaire, is one of the most frequently used instruments to measure self-recall of physical activity in cancer survivors (Amireault et al., 2015b). This succinct four-item questionnaire is recommended by the National Cancer Institute Division of Cancer Epidemiology and Genetics research program for oncology clinicians and researchers. The GSLTPAQ ranks survivors as either active (score of 24 or higher) or inactive (i.e., insufficiently active) (score of 23 or lower) on a validated leisure score index. Correlations with physical activity as measured by accelerometer or pedometer range from 0.31 to 0.57, and agreement between GSLTPAQ scores and accelerometer measurement was 70.8% in a sample of 199 breast cancer survivors (Amireault et al., 2015a, 2015b).
PSQI: The PSQI is a validated instrument commonly used for measuring sleep quality in cancer-related studies. A study with 474 breast cancer survivors demonstrated strong internal consistency (Cronbach’s alpha = 0.7), test–retest reliability (intraclass correlation coefficient = 0.76), and correlation with subjective sleep complaints (r ≥ 0.60) (Fontes et al., 2017). The PSQI contains seven component scores calculated from 18 items and yields a global score. Higher scores indicate greater difficulty sleeping.
Mindful Attention Awareness Scale: The Mindful Attention Awareness Scale is a six-point, 15-item Likert-type scale with scores ranging from 1 (almost always) to 6 (almost never). Higher scores indicate higher levels of mindfulness (Brown & Ryan, 2003). The instrument is used to assess the core characteristic of mindfulness and has good internal consistency (Cronbach’s alpha = 0.82–0.87) and divergent validity with the Beck Depression Inventory and State–Trait Anxiety Inventory (Dehghan et al., 2020).
PROMIS Emotional Distress–Anxiety and PROMIS Emotional Distress–Depression Short Forms: These PROMIS instruments are four-item, five-point Likert-type scales with scores ranging from 1 (never) to 5 (always). Higher scores indicate greater anxiety or depression. Raw scores are rescaled into T scores with a mean of 50 and an SD of 10. These instruments have been extensively validated in multiple populations, including cancer survivors. Individual PROMIS negative affect domains for anxiety and depression were validated with more than 300 cancer survivors in addition to other populations. Validity of these subscales was found to be excellent (Cronbach’s alpha = 0.97) (Schalet et al., 2016).
UCLA Loneliness Scale: This instrument has been used extensively in the cancer survivor population to assess subjective feelings of loneliness and isolation. The UCLA Loneliness Scale, version 3.0, consists of 20 items ranking loneliness from 1 (never) to 4 (often) and yields a total score, with higher total scores indicating greater self-reported loneliness (Russell, 1996). Internal consistency reports are high, with Cronbach’s alpha ranging from 0.89 to 0.94 and test–retest reliability during a 12-month period reported as r = 0.73 (Deckx et al., 2014).
Determinants of behavior change (intention): This researcher-designed measure was developed according to established procedures for the design of questionnaires based on the TPB (Ajzen, 1991, 2006). Intention to change was selected because it is the most proximal determinant of behavior change according to the TPB. Two items are ranked on a seven-point Likert-type scale. The scores of both items are summed to yield the intentions construct score. Test–retest analysis in a previous single-arm pilot study found high reliability (Cronbach’s alpha ≥ 0.7) across three behaviors (exercise, sleep, and mindfulness) (Myers et al., 2022).
PROMIS Global Health: This 10-item measure of HRQOL yields global physical and mental health scores and is scored in the same manner as other PROMIS instruments. Internal consistency (0.81) and reliability (0.86) are excellent (Hays et al., 2009).
Satisfaction survey: This 20-item survey was created by the researchers and used to assess participants’ satisfaction with the length of the intervention, class session duration, and weekly program content. Items were scored from 0 (not satisfied at all) to 4 (exceptionally satisfied). Participants also were asked to rank their satisfaction with the homework assignments, number and length of study questionnaires, and completion of study questionnaires online, as well as to indicate their preferred method for Haze delivery if they were to take part in the Haze program again. Three narrative response questions were included to gather qualitative data about aspects of the Haze program that participants found to be most helpful, strategies gleaned from the program they planned to use regularly, and any other feedback they wished to share with the team about the Haze program or the study experience. This instrument was administered only at T2, and these data were not collected from participants in the WLC group.
Statistical Analyses
Descriptive statistics (counts, means, and SDs) were used to summarize results for aim 1 (feasibility measures: enrollment, retention, and satisfaction), and within- and between-group differences for study outcomes at T2 and T3 were used for aim 2 (primary and secondary measures of PCF) and aim 3 (changes in health behaviors, psychosocial outcomes, intention to change, and HRQOL). Linear mixed models also were used to test the within-group changes (T3 versus T2) and between-group differences in changes in study outcomes for aims 2 and 3. The following potential confounding factors were controlled in these models: baseline study questionnaire scores, age, years of education, and total types of treatment. Two contrasts of interest were tested. One contrast was used to compare differences in scores between the WLC and the two intervention groups combined; the other contrast was used to compare differences in scores between the live and prerecorded groups. Because all analyses were conducted based on changes in scores between time points, only participants for whom data were available for all time points were included in the between-group comparisons of changes. Results of the analyses were compared using the last observation carried forward missing data computation method. Consistency between these results confirmed the robustness of the findings.
Participants’ responses to the narrative questions on the satisfaction survey were reviewed using deductive content analysis with the goal of gaining feedback to inform future development of the intervention. Themes were developed based on the satisfaction survey items to describe aspects of the Haze program that participants found to be most helpful, as well as any action plans for implementation of strategies learned from Haze content.
Results
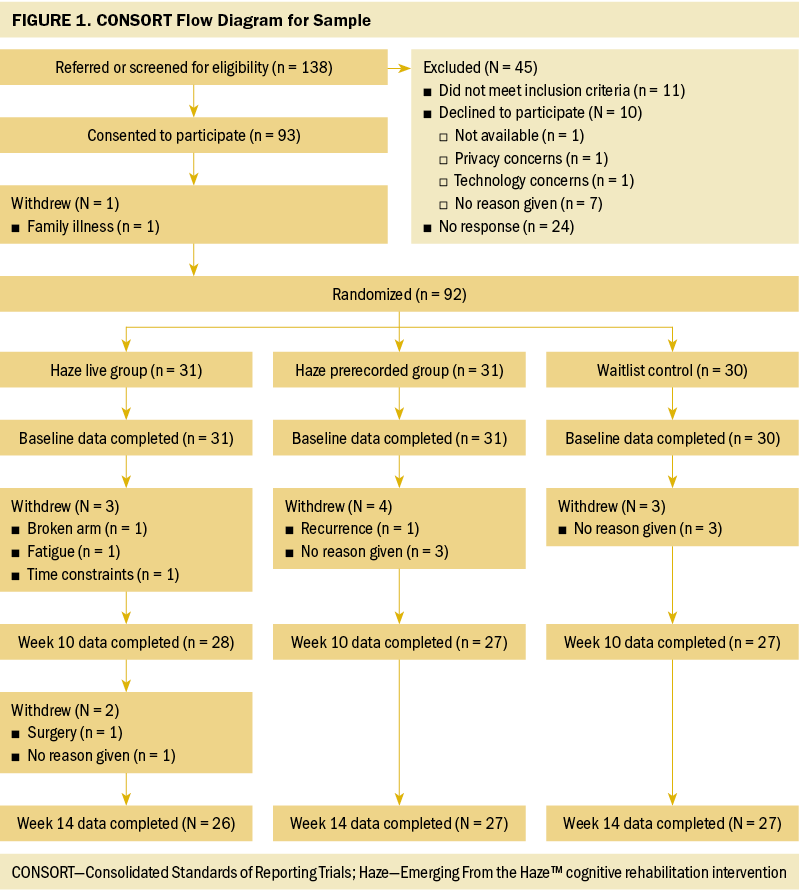
One hundred and thirty-eight patients were referred and/or screened for participation (see Figure 1). Target enrollment (N = 90) was exceeded; 93 participants provided consent. Participants were geographically diverse, with representation across three time zones (Pacific, Central, and Eastern). One participant withdrew prior to completion of the baseline questionnaires and randomization, providing an initial evaluable sample of 92 participants. Participants primarily were White (n = 73), non-Hispanic (n = 81), female (n = 89), and diagnosed with breast cancer (n = 64) (see Table 2). The majority were married (n = 56) and well educated (mean years of education = 16.33). Slightly more than half (n = 51) had received radiation therapy. About one-third (n = 28) had received endocrine therapy, 25 of whom were receiving this treatment at the time of the study. No significant demographic differences were noted between groups at baseline. However, baseline scores on the PROMIS CF and CF-A instruments measuring PCF were higher for both intervention groups than for the WLC (p < 0.05).
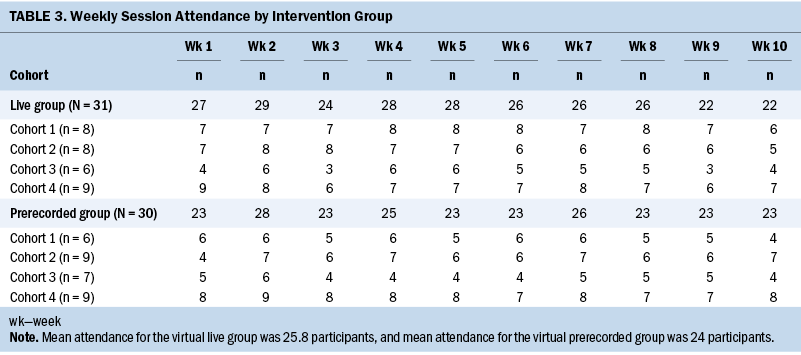
Weekly attendance for each cohort is reflected by group in Table 3. Mean weekly attendance was 25.8 of 31 for the live group and 24 of 31 for the prerecorded group. Overall retention was 82 of 93, with 26 of 31 for the live group, 27 of 31 for the prerecorded group, and 27 of 30 for the WLC.
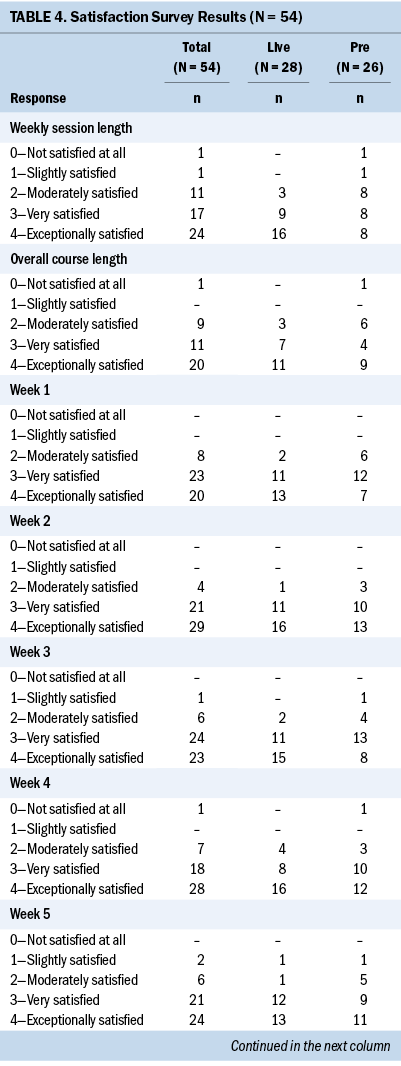
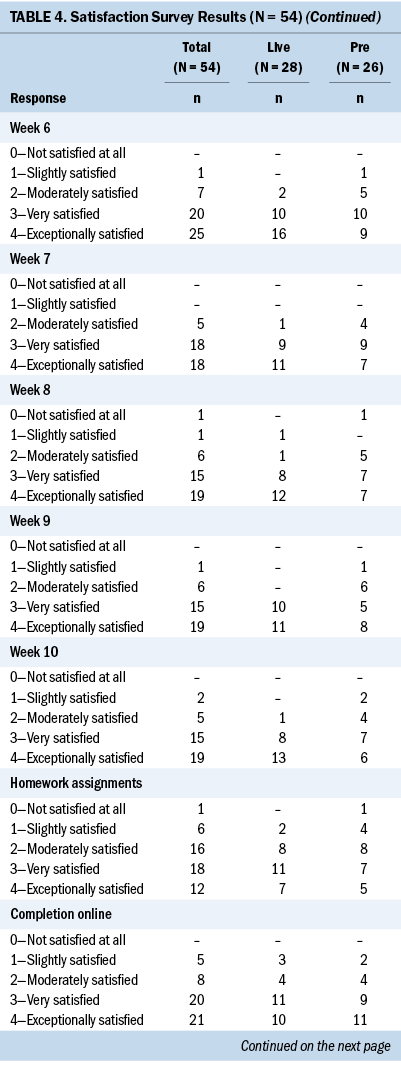
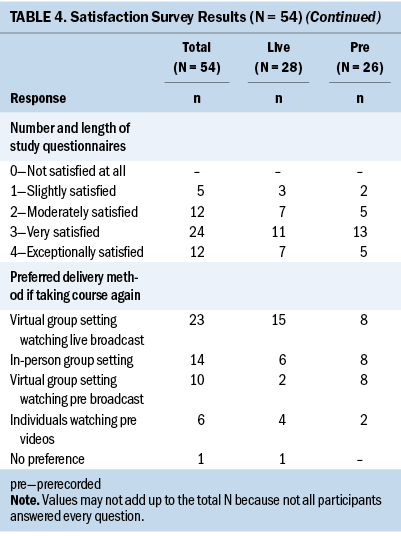
Participant satisfaction ratings for length of the intervention, class session duration, and weekly program content after completion of the live and prerecorded group Haze sessions were high (see Table 4). Across 10 weekly sessions, overall satisfaction ratings of 2 or higher on a 0–4 scale ranged from 83% to 100% (mean = 95%), and satisfaction ratings of 3 or higher ranged from 76% to 93% (mean = 83%). Similar results were noted for the completion of study questionnaires online. Average satisfaction ratings across 10 weekly sessions of 2 or higher were 91%, and satisfaction ratings of 3 or higher were 76%. Satisfaction ratings with the homework assignments and number and length of study questionnaires were slightly lower. Across these two items, ratings of 2 or higher ranged from 87% to 91%, and ratings of 3 or higher ranged from 57% to 68%. Satisfaction ratings for the live and prerecorded groups were comparable across all the measures. Participants were asked to indicate their preferred method of Haze delivery if they were to take part in the program again. Of 54 participants, 23 preferred the virtual live group, 14 preferred an in-person group setting, 10 preferred the virtual prerecorded group, 6 preferred individual viewing, and 1 expressed no preference. When the responses were calculated by intervention group, 15 of 28 participants in the virtual live group indicated that they would choose that delivery method again. Participants in the virtual prerecorded group were evenly distributed at 8 of 26 each for choosing in-person, live, and prerecorded delivery if they were to take part in the program again.
Narrative responses on the satisfaction survey were provided by 43 participants. Of the 28 who responded to the question about what components of the Haze program they planned to implement, the most common answer was mindfulness (n = 28), followed by diet (n = 19), exercise (n = 18), sleep (n = 17), and experiential strategies to enhance memory and concentration (n = 14).

Positive comments were provided about the strength of the presenters, empathy of the group facilitator, and experience of feeling heard. Participants in both intervention groups mentioned appreciating the ability to ask questions about the Haze content. Several participants mentioned enjoying the group interaction and the validation they experienced from knowing they were not alone regarding the experience of CRCI. Direct quotes demonstrating participants’ descriptions of what they found most valuable about the Haze program and what they intend to implement are listed in Figure 2.
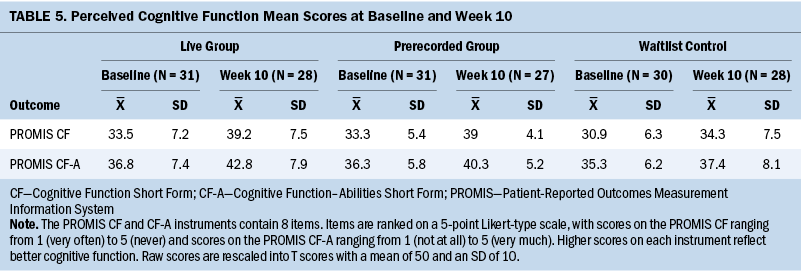
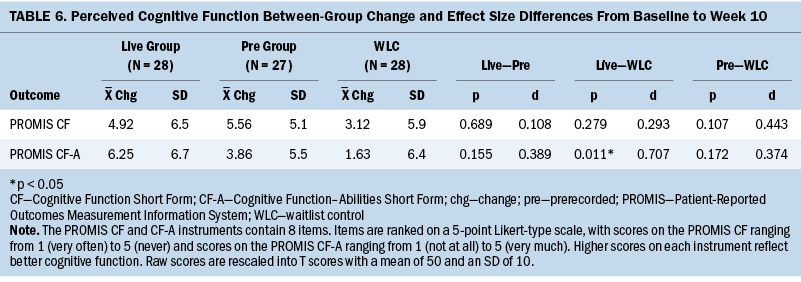
Descriptive statistics (means and SDs) for the variables used to measure PCF (PROMIS CF and CF-A) are listed in Table 5. Two-sample t tests indicated that the only statistically significant between-group difference demonstrated was in PROMIS CF-A scores for the virtual live group compared to WLC at T2 (p = 0.011, d = 0.707) (see Table 6). No differences were seen in the CF (p = 0.689, d = 0.108) or CF-A (p = 0.155, d = 0.389) scores between the live and prerecorded groups. Clinically meaningful improvement in the CF-A score was demonstrated for the prerecorded group (p = 0.107, d = 0.413) between T1 and T2 compared to WLC in congruence with the minimally important change defined for PROMIS measures (between 2 and 6 T scores).
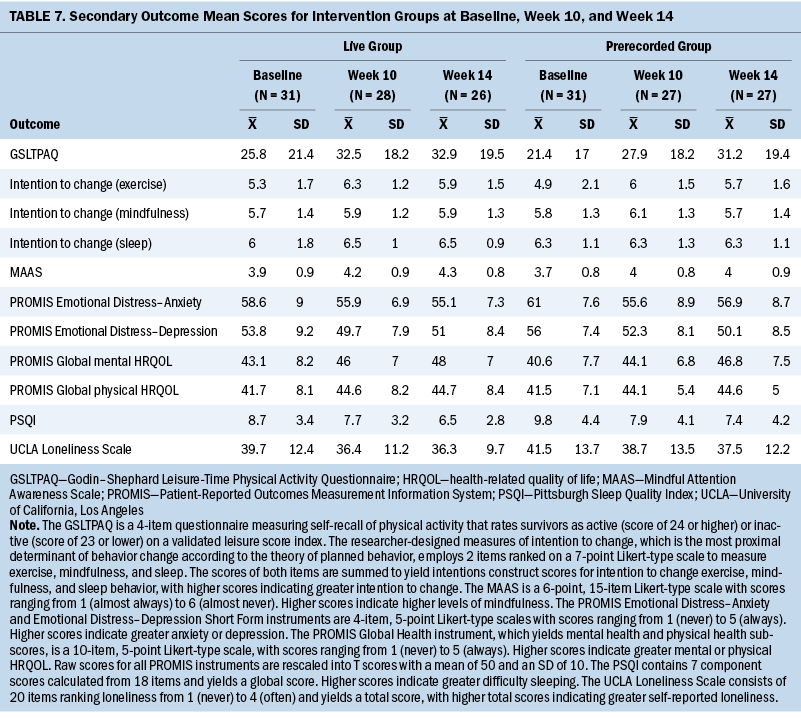
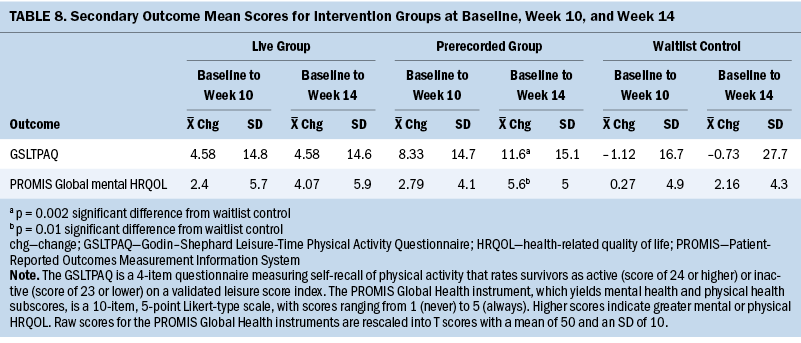
Descriptive statistics (means and SDs) for the measures of health behaviors, psychosocial outcomes, determinants of behavior change, and HRQOL are listed in Table 7. Two-sample t tests or Wilcoxon rank sum tests were conducted to compare the between-group difference in change from baseline for each outcome depending on whether the normality of data assumption was satisfied. The only significant between-group change scores were demonstrated between T1 and T3 for the prerecorded group compared to the WLC. The mean change in physical activity (GSLTPAQ scores) for the prerecorded group was 11.6 (SD = 15.1) compared to a mean change of –0.73 (SD = 27.7) for the WLC (p = 0.002) (see Table 8). In addition, an improvement in mental HRQOL (PROMIS Global Health score) was demonstrated between T1 and T3 for the prerecorded group (mean change = 5.6, SD = 5) compared to the WLC (mean change = 2.16, SD = 4.3) (p = 0.01).
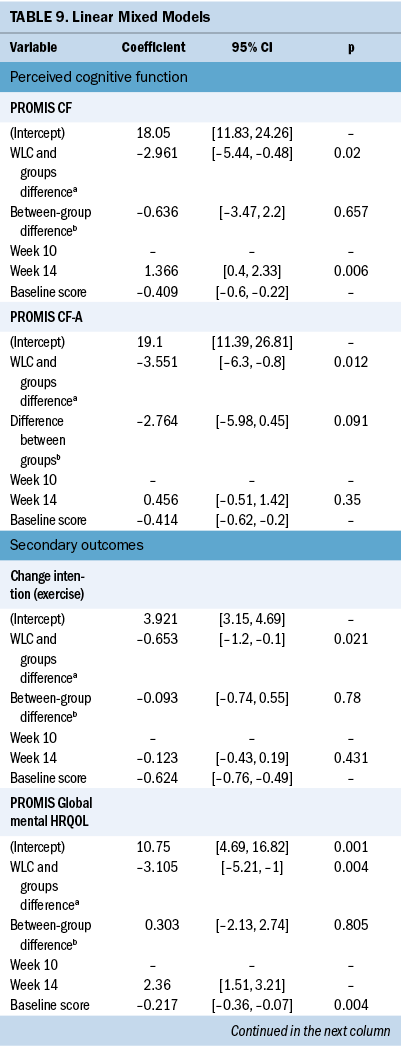
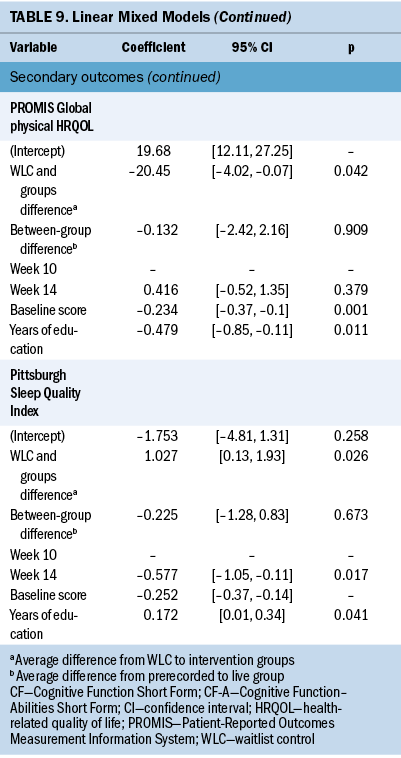
Because of the lack of between-group difference for the live and prerecorded groups, linear mixed models (random intercept) were used to analyze changes in scores between time points (T2–T1 and T3–T1) for repeated measures after controlling for baseline scores, within-group effects (T3 versus T2), and between-group effects (randomized group). Univariate analyses of associations of potential confounding factors (age, years of education, total types of treatment) for each outcome were performed prior to conducting linear mixed-model analyses. Significant results are reported in Table 9. Significant modeling estimates for the two intervention groups combined were obtained for PCF (PROMIS CF and CF-A, higher scores indicate better cognition), sleep quality (PSQI, lower scores indicate better sleep quality), intention to change exercise behavior, and physical and mental HRQOL (PROMIS Global Health, higher scores indicate greater HRQOL).
Discussion
The original Haze intervention was delivered in person during six weekly 2.5-hour sessions (Myers et al., 2020). Following the onset of the COVID-19 pandemic, the Haze intervention was piloted live via secure videoconferencing (Myers et al., 2022). Although satisfaction survey results did not indicate the need for change, feedback gleaned from interviews conducted with participants in the earlier pilot informed the revision of the Haze intervention from six weekly 2.5-hour sessions to 10 weekly 60- to 90-minute sessions to minimize “Zoom fatigue” (Myers et al., 2022). Results from the current three-arm pilot study demonstrated that the shift to a 10-week duration and shorter weekly sessions was acceptable to participants. Intervention group participants rated satisfaction with overall course length and weekly session length as “very satisfied” (17 of 54 for overall course length, 18 of 54 for weekly session length) or “exceptionally satisfied” (both 24 of 54). Satisfaction with the weekly session content also was high. On average across 10 weeks, slightly more than 84% rated satisfaction with weekly session content (weekly sessions were attended by differing numbers of participants) as “very satisfied” (39%) or “exceptionally satisfied” (45%). Only 14 of 54 participants indicated a preference for in-person group Haze delivery if they were to take part in the program again. Satisfaction with completing the study questionnaires online also was high, with the majority (41 of 54) having reported being “very” or “exceptionally” satisfied. These results appear to reflect participants’ comfort with the shift to virtual programming and research participation.
Overall participant retention was high (n = 82 of 93), as was retention in the live group (n = 26 of 31) and prerecorded group (n = 27 of 31). As with previous studies, the researchers believe that the WLC design contributed to the retention rate for the WLC group (n = 27 of 30) in addition to having a positive influence on recruitment because all participants were able to eventually attend the Haze program. Additional elements of feasibility were demonstrated through successful recruitment of the desired sample size and weekly session attendance ranging from 22 to 29 of 31 for the live group (mean = 25.8 of 31) and 23 to 26 of 30 for the prerecorded group (mean = 24 of 30) (some group participants dropped out of the study over time, affecting weekly attendance rates).
Very few participants required the loaned cellular data plan–enabled tablets (n = 3). However, this pilot study provided the opportunity to further develop and test a system for provision and return of the tablets to inform future work. Of note, referring healthcare providers shared that the availability of the tablets contributed to being able to offer the program to patients living in rural areas and patients without internet access. This practice should be continued in future research to facilitate efforts to increase sample diversity. Participants in this pilot primarily were non-Hispanic (n = 81) and White (n = 73). Efforts are ongoing to translate the Haze intervention and related materials for Spanish-speaking participants. These revisions will facilitate future targeted recruitment among the Hispanic population.
In addition to meeting the primary end points for feasibility, the study results contributed to the growing body of evidence in support of cognitive rehabilitation interventions to address CRCI for cancer survivors. Improvements in PCF, as measured by the PROMIS CF and CF-A, were demonstrated for both intervention groups. Statistically significant difference in the PROMIS CF-A score between the live group and WLC was observed (p = 0.011). In addition, clinically meaningful difference in the PROMIS CF-A score was demonstrated for the prerecorded group compared to the WLC (Cohen’s d = 0.443), which supports findings from earlier pilot studies (Myers et al., 2020; Terwee et al., 2021). However, no between-group differences were noted for the two intervention groups. Improvements were observed for sleep quality, intention to change exercise behavior, and physical and mental HRQOL in the intervention groups compared to WLC. In contrast to previous studies, reduction in loneliness did not reach statistical significance for the intervention groups (Myers et al., 2020, 2022). One potential explanation may be that participants had developed effective strategies for coping with loneliness and isolation during the COVID-19 pandemic. Another explanation may be that virtual programming is not a consistently ideal way to mitigate loneliness.
Limitations
Study limitations include the relatively small numbers of participants for each of the three groups. The study was powered to detect only a large effect size, and this limitation may have blunted the detection of between-group differences. Despite broad geographic representation, participant racial and ethnic diversity was limited primarily to White, non-Hispanic survivors, minimizing generalizability of results to a broader population. The fact that some participants were recruited from the Haze waitlist at CSMC may have more heavily weighted participant distribution to the West Coast, but the researchers do not believe that this distribution had an impact on feasibility or participant satisfaction. Patient-reported outcomes were chosen as the primary measure of cognitive function and abilities for this pilot study. Evidence would suggest that subjective (patient-reported) and objective (neurocognitive testing) measures of cognitive function may measure different constructs, and the psychological impact of CRCI may be most associated with effects on HRQOL (Gutenkunst et al., 2021; Hermelink et al., 2010; Oerlemans et al., 2022). The sensitivity of neurocognitive testing for non–central nervous system CRCI remains a subject of some debate, particularly with relatively short intervals of time before and after an intervention and related to ecologic validity shortfalls germane to several psychometric measures. The search for accessible, undemanding testing to evaluate day-to-day neurocognitive performance in a nonclinical testing environment continues (Savard & Ganz, 2016). Results from a concurrently conducted substudy of a mobile digital assessment tool will be reported separately.
Findings from the satisfaction survey were limited because feedback was gleaned from participants who had experienced only a single Haze delivery method. Those who were in the virtual live group indicated a preference for virtual live group delivery again if they were to repeat the Haze program. Interestingly, those in the prerecorded group indicated equal preference among in-person, virtual live, and virtual prerecorded group delivery. Having a positive experience with the prerecorded delivery method, as evidenced by the high satisfaction ratings, may have influenced responses to this question. The flexibility for dissemination and scheduling provided by the prerecorded Haze delivery format, combined with the lack of between-group differences in outcomes from the live group, warrant additional study in a larger, well-powered, randomized study. Of note, during recruitment, some potential and actual participants expressed the desire for individual viewing of the prerecorded content. This option was requested because of timing conflicts between work schedules and the study cohorts (day of week, time of day), as well as because some participants wanted to “make up” content that they missed because of being absent during the 10 weekly sessions. Of the 54 intervention participants, 6 indicated that individual viewing would be their preferred option if they were to participate in the program again. Future research is needed to compare virtual delivery in a group setting and virtual delivery to individuals.
The goal for future development and testing of the prerecorded version will be to increase the strength and ability of the intervention to be broadly disseminated to a wider range of geographically diverse and under-resourced populations. The current pilot was conducted by members of the study team to lay the necessary groundwork for larger-scale testing with multiple group facilitators across multiple settings.
Implications for Nursing
CRCI is a prevalent sequela to cancer and cancer treatment and has a significant negative impact on HRQOL for cancer survivors. The results of this study add to the evidence in support of cognitive rehabilitation programs to address the cognitive issues reported by survivors and therapeutic lifestyle interventions. Cognitive rehabilitation programs blend experiential exercises to improve memory and concentration with strategies for coping with changes in cognitive function. Nurses play a key role in identifying patients who are experiencing these changes. Nurses’ awareness of available cognitive rehabilitation programs and eligibility requirements for ongoing clinical trials is critical for providing cancer survivors with access to appropriate care.

Conclusion
Limited resources are available to meet the rehabilitation needs of the broader community, and numerous barriers still exist for individuals with functional difficulties or disabilities that interfere with access to rehabilitative interventions (Bright et al., 2018). Demonstrating feasibility (recruitment, retention, and satisfaction) for two forms of telehealth virtual delivery (live versus prerecorded) of the Haze program compared to WLC was a critical step to inform the design of a larger confirmatory study, which could demonstrate the evidence necessary to support the program’s accessibility and wide dissemination to a broad and underserved population of cancer survivors. An important next step toward this goal will be to test implementation of the prerecorded version across multiple sites and group facilitators to test feasibility and intervention fidelity on a broader scale in preparation for a large confirmatory trial. Future research is needed to examine the potential impact of the prerecorded cognitive rehabilitation content that may be viewed individually (i.e., outside of a group setting). Culturally appropriate translations for non–English-speaking participants also are of interest to facilitate health equity for survivors experiencing CRCI.
The authors gratefully acknowledge David Streeter for his support as director of cancer informatics and the Curated Cancer Clinical Outcomes Database, which was used to inform recruitment strategies for study participants from the University of Kansas Cancer Center.
About the Authors
Jamie S. Myers, PhD, RN, AOCNS®, FAAN, is a research associate professor in the School of Nursing at the University of Kansas in Kansas City; Jeffrey C. Wertheimer, PhD, ABPP-CN, FACRM, is the associate director of physical medicine and rehabilitation and chief of psychology and neuropsychology services at Cedars-Sinai Medical Center in Los Angeles, CA; Jaromme Geebum Kim, MA, is a graduate research assistant and Jianghua He, PhD, is a professor, both in the Department of Biostatistics and Data Science at the University of Kansas Medical Center in Kansas City; Celina H. Shirazipour, PhD, MHK, BA, is a research scientist at Cedars-Sinai Medical Center; Dedrick Hooper, BS, is a senior systems coordinator in the Center for Telemedicine and Telehealth, Mary Penne Mays, BS, is a clinical informatics coordinator in the Office of the Chief Research Informatics Officer, and Jennifer R. Klemp, PhD, MPH, is a co–program leader of cancer prevention and control and a professor of medicine in the Division of Medical Oncology, all at the University of Kansas Medical Center; Carli Zegers, PhD, MBA, APRN, FNP-BC™, is an associate professor in the College of Nursing at Florida State University in Tallahassee; and Arash Asher, MD, is an associate professor of physical medicine and rehabilitation and director of cancer rehabilitation and survivorship at Cedars-Sinai Medical Center. This research was funded, in part, by an Oncology Nursing Foundation endowment and through a contribution made to the Foundation from Genentech. Myers, Wertheimer, He, Shirazipour, Penne Mays, Klemp, and Asher contributed to the conceptualization and design. Myers, Wertheimer, Penne Mays, Klemp, and Asher completed the data collection. Myers, Kim, He, Hooper, and Asher provided statistical support. Myers, Wertheimer, He, Klemp, and Asher provided the analysis. Myers, Wertheimer, He, Shirazipour, Zegers, and Asher contributed to the manuscript preparation. Myers can be reached at jmyers@kumc.edu, with copy to ONFEditor@ons.org. (Submitted November 2023. Accepted February 5, 2024.)
References
Ahles, T.A., & Root, J.C. (2018). Cognitive effects of cancer and cancer treatments. Annual Review of Clinical Psychology, 14, 425–451. https://doi.org/10.1146/annurev-clinpsy-050817-084903
Ahles, T.A., Root, J.C., & Ryan, E.L. (2012). Cancer- and cancer treatment–associated cognitive change: An update on the state of the science. Journal of Clinical Oncology, 30(30), 3675–3686. https://doi.org/10.1200/JCO.2012.43.0116
Ahles, T.A., & Saykin, A.J. (2007). Candidate mechanisms for chemotherapy-induced cognitive changes. Nature Reviews Cancer, 7(3), 192–201. https://doi.org/10.1038/nrc2073
Ajzen, I. (1991). The theory of planned behavior. Organizational Behavior and Human Decision Processes, 50(2), 179–211. https://doi.org/10.1016/0749-5978(91)90020-T
Ajzen, I. (2006). Constructing a theory of planned behavior questionnaire: Conceptual and methodological considerations. https://people.umass.edu/aizen/pdf/tpb.measurement.pdf
Ajzen, I., & Fishbein, M. (1980). Understanding attitudes and predicting social behavior. Prentice-Hall.
Allen, D.H., Myers, J.S., Jansen, C.E., Merriman, J.D., & Von Ah, D. (2018). Assessment and management of cancer- and cancer treatment–related cognitive impairment. Journal for Nurse Practitioners, 14(4), 217–224. https://doi.org/10.1016/j.nurpra.2017.11.026
Amireault, S., Godin, G., Lacombe, J., & Sabiston, C.M. (2015a). The use of the Godin–Shephard Leisure-Time Physical Activity Questionnaire in oncology research: A systematic review. BMC Medical Research Methodology, 15, 60. https://doi.org/10.1186/s12874-015-0045-7
Amireault, S., Godin, G., Lacombe, J., & Sabiston, C.M. (2015b). Validation of the Godin–Shephard Leisure-Time Physical Activity Questionnaire classification coding system using accelerometer assessment among breast cancer survivors. Journal of Cancer Survivorship, 9(3), 532–540. https://doi.org/10.1007/s11764-015-0430-6
Asher, A., & Myers, J.S. (2015). The effect of cancer treatment on cognitive function. Clinical Advances in Hematology and Oncology, 13(7), 441–450.
Asher, A., Van Dyk, K., Jo, M.-Y., Bailey, C., & Myers, J.S. (2019). Cancer-related cognitive impairment: Retrospective analyses of a multidimensional, psychoeducation-based cognitive rehabilitation intervention. Clinical Journal of Oncology Nursing, 23(3), 301–308. https://doi.org/10.1188/19.CJON.301-308
Boykoff, N., Moieni, M., & Subramanian, S.K. (2009). Confronting chemobrain: An in-depth look at survivors’ reports of impact on work, social networks, and health care response. Journal of Cancer Survivorship, 3(4), 223–232. https://doi.org/10.1007/s11764-009-0098-x
Bright, T., Wallace, S., & Kuper, H. (2018). A systematic review of access to rehabilitation for people with disabilities in low- and middle-income countries. International Journal of Environmental Research and Public Health, 15(10), 2165. https://doi.org/10.3390/ijerph15102165
Brown, K.W., & Ryan, R.M. (2003). The benefits of being present: Mindfulness and its role in psychological well-being. Journal of Personality and Social Psychology, 84(4), 822–848. https://doi.org/10.1037/0022-3514.84.4.822
Browne, R.H. (1995). On the use of a pilot sample for sample size determination. Statistics in Medicine, 14(17), 1933–1940. https://doi.org/10.1002/sim.4780141709
Cacioppo, J.T., & Hawkley, L.C. (2009). Perceived social isolation and cognition. Trends in Cognitive Sciences, 13(10), 447–454. https://doi.org/10.1016/j.tics.2009.06.005
Chiesa, A., Calati, R., & Serretti, A. (2011). Does mindfulness training improve cognitive abilities? A systematic review of neuropsychological findings. Clinical Psychology Review, 31(3), 449–464. https://doi.org/10.1016/j.cpr.2010.11.003
Crouch, A., Champion, V., & Von Ah, D. (2022). Cognitive dysfunction in older breast cancer survivors: An integrative review. Cancer Nursing, 45(1), E162–E178. https://doi.org/10.1097/ncc.0000000000000896
Deckx, L., van den Akker, M., & Buntinx, F. (2014). Risk factors for loneliness in patients with cancer: A systematic literature review and meta-analysis. European Journal of Oncology Nursing, 18(5), 466–477. https://doi.org/10.1016/j.ejon.2014.05.002
Dehghan, M., Jazinizade, M., Malakoutikhah, A., Madadimahani, A., Iranmanesh, M.H., Oghabian, S., . . . Zakeri, M.A. (2020). Stress and quality of life of patients with cancer: The mediating role of mindfulness. Journal of Oncology, 2020, 3289521. https://doi.org/10.1155/2020/3289521
Di Liegro, C.M., Schiera, G., Proia, P., & Di Liegro, I. (2019). Physical activity and brain health. Genes, 10(9), 720. https://doi.org/10.3390/genes10090720
Dos Santos, M., Hardy-Léger, I., Rigal, O., Licaj, I., Dauchy, S., Levy, C., . . . Joly, F. (2020). Cognitive rehabilitation program to improve cognition of cancer patients treated with chemotherapy: A 3-arm randomized trial. Cancer, 126(24), 5328–5336. https://doi.org/10.1002/cncr.33186
Fernandes, H.A., Richard, N.M., & Edelstein, K. (2019). Cognitive rehabilitation for cancer-related cognitive dysfunction: A systematic review. Supportive Care in Cancer, 27(9), 3253–3279. https://doi.org/10.1007/s00520-019-04866-2
Fontes, F., Gonçalves, M., Maia, S., Pereira, S., Severo, M., & Lunet, N. (2017). Reliability and validity of the Pittsburgh Sleep Quality Index in breast cancer patients. Supportive Care in Cancer, 25(10), 3059–3066. https://doi.org/10.1007/s00520-017-3713-9
Gutenkunst, S.L., Vardy, J.L., Dhillon, H.M., & Bell, M.L. (2021). Correlates of cognitive impairment in adult cancer survivors who have received chemotherapy and report cognitive problems. Supportive Care in Cancer, 29(3), 1377–1386. https://doi.org/10.1007/s00520-020-05616-5
Harris, P.A., Taylor, R., Minor, B.L., Elliott, V., Fernandez, M., O’Neal, L., . . . Duda, S.N. (2019). The REDCap consortium: Building an international community of software platform partners. Journal of Biomedical Informatics, 95, 103208. https://doi.org/10.1016/j.jbi.2019.103208
Harris, P.A., Taylor, R., Thielke, R., Payne, J., Gonzalez, N., & Conde, J.G. (2009). Research Electronic Data Capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. Journal of Biomedical Informatics, 42(2), 377–381. https://doi.org/10.1016/j.jbi.2008.08.010
Hays, R.D., Bjorner, J.B., Revicki, D.A., Spritzer, K.L., & Cella, D. (2009). Development of physical and mental health summary scores from the Patient-Reported Outcomes Measurement Information System (PROMIS) Global items. Quality of Life Research, 18(7), 873–880. https://doi.org/10.1007/s11136-009-9496-9
Henderson, F.M.E., Cross, A.J., & Baraniak, A.R. (2019). ‘A new normal with chemobrain’: Experiences of the impact of chemotherapy-related cognitive deficits in long-term breast cancer survivors. Health Psychology Open, 6(1), 2055102919832234. https://doi.org/10.1177/2055102919832234
Henneghan, A.M., Van Dyk, K., Kaufmann, T., Harrison, R., Gibbons, C., Heijnen, C., & Kesler, S.R. (2021). Measuring self-reported cancer-related cognitive impairment: Recommendations from the Cancer Neuroscience Initiative Working Group. Journal of the National Cancer Institute, 113(12), 1625–1633. https://doi.org/10.1093/jnci/djab027
Henneghan, A.M., Van Dyk, K., Zhou, X., Moore, R.C., Root, J.C., Ahles, T.A., . . . Ganz, P.A. (2023). Validating the PROMIS Cognitive Function Short Form in cancer survivors. Breast Cancer Research and Treatment, 201(1), 139–145. https://doi.org/10.1007/s10549-023-06968-2
Hermelink, K., Küchenhoff, H., Untch, M., Bauerfeind, I., Lux, M.P., Bühner, M., . . . Münzel, K. (2010). Two different sides of ‘chemobrain’: Determinants and nondeterminants of self-perceived cognitive dysfunction in a propspective, randomized, multicenter study. Psycho-Oncology, 19(12), 1321–1328. https://doi.org/10.1002/pon.1695
Hess, L.M., & Insel, K.C. (2007). Chemotherapy-related change in cognitive function: A conceptual model. Oncology Nursing Forum, 34(5), 981–994. https://doi.org/10.1188/07.ONF.981-994
Janelsins, M.C., Kesler, S.R., Ahles, T.A., & Morrow, G.R. (2014). Prevalence, mechanisms, and management of cancer-related cognitive impairment. International Review of Psychiatry, 26(1), 102–113. https://doi.org/10.3109/09540261.2013.864260
Jaremka, L.M., Peng, J., Bornstein, R., Alfano, C.M., Andridge, R.R., Povoski, S.P., . . . Kiecolt-Glaser, J.K. (2014). Cognitive problems among breast cancer survivors: Loneliness enhances risk. Psycho-Oncology, 23(12), 1356–1364. https://doi.org/10.1002/pon.3544
Johns, S.A., Brown, L.F., Beck-Coon, K., Talib, T.L., Monahan, P.O., Giesler, R.B., . . . Kroenke, K. (2016). Randomized controlled pilot trial of mindfulness-based stress reduction compared to psychoeducational support for persistently fatigued breast and colorectal cancer survivors. Supportive Care in Cancer, 24(10), 4085–4096. https://doi.org/10.1007/s00520-016-3220-4
Julious, S.A. (2005). Sample size of 12 per group rule of thumb for a pilot study. Pharmaceutical Statistics, 4(4), 287–291. https://doi.org/10.1002/pst.185
Koppelmans, V., de Groot, M., de Ruiter, M.B., Boogerd, W., Seynaeve, C., Vernooij, M.W., . . . Breteler, M.M.B. (2014). Global and focal white matter integrity in breast cancer survivors 20 years after adjuvant chemotherapy. Human Brain Mapping, 35(3), 889–899. https://doi.org/10.1002/hbm.22221
Lai, J.-S., Wagner, L.I., Jacobsen, P.B., & Cella, D. (2014). Self-reported cognitive concerns and abilities: Two sides of one coin? Psycho-Oncology, 23(10), 1133–1141. https://doi.org/10.1002/pon.3522
Lange, M., Joly, F., Vardy, J., Ahles, T., Dubois, M., Tron, L., . . . Castel, H. (2019). Cancer-related cognitive impairment: An update on state of the art, detection, and management strategies in cancer survivors. Annals of Oncology, 30(12), 1925–1940. https://doi.org/10.1093/annonc/mdz410
Liang, M.I., Erich, B., Bailey, C., Jo, M.-Y., Walsh, C.S., & Asher, A. (2018). Emerging From the Haze: A pilot study evaluating feasibility of a psychoeducational intervention to improve cancer-related cognitive impairment in gynecologic cancer survivors. Journal of Palliative Care, 34(1), 32–37. https://doi.org/10.1177/0825859718796794
Myers, J.S. (2009). A comparison of the theory of unpleasant symptoms and the Conceptual Model of Chemotherapy-Related Changes in Cognitive Function. Oncology Nursing Forum, 36, E1–E10. https://doi.org/10.1188/09.ONF.E1-E10
Myers, J.S. (2012). Chemotherapy-related cognitive impairment: The breast cancer experience. Oncology Nursing Forum, 39(1), E31–E40. https://doi.org/10.1188/12.ONF.E31-E40
Myers, J.S., Cook-Wiens, G., Baynes, R., Jo, M.-Y., Bailey, C., Krigel, S., . . . Asher, A. (2020). Emerging From the Haze: A multicenter, controlled pilot study of a multidimensional, psychoeducation-based cognitive rehabilitation intervention for breast cancer survivors delivered with telehealth conferencing. Archives of Physical Medicine and Rehabilitation, 101(6), 948–959. https://doi.org/10.1016/j.apmr.2020.01.021
Myers, J.S., Shirazipour, C.H., Wertheimer, J.C., & Asher, A. (2022). Feasibility pilot study of a virtual intervention for survivors with decreased perceived cognitive function after cancer treatment. Oncology Nursing Forum, 49(1), 90–95. https://doi.org/10.1188/22.ONF.90-95
National Comprehensive Cancer Network. (2023). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Survivorship [v.1.2023]. https://www.nccn.org
Oerlemans, S., Schagen, S.B., van den Hurk, C.J., Husson, O., Schoormans, D., & van de Poll-Franse, L.V. (2022). Self-perceived cognitive functioning and quality of life among cancer survivors: Results from the PROFILES registry. Journal of Cancer Survivorship, 16(2), 303–313. https://doi.org/10.1007/s11764-021-01023-9
Rhodes, R.E., & Courneya, K.S. (2004). Differentiating motivation and control in the Theory of Planned Behavior. Psychology, Health and Medicine, 9(2), 205–215. https://doi.org/10.1080/13548500410001670726
Russell, D.W. (1996). UCLA Loneliness Scale (version 3): Reliability, validity, and factor structure. Journal of Personality Assessment, 66(1), 20–40. https://doi.org/10.1207/s15327752jpa6601_2
Savard, J., & Ganz, P.A. (2016). Subjective or objective measures of cognitive functioning—What’s more important? JAMA Oncology, 2(10), 1263–1264. https://doi.org/10.1001/jamaoncol.2016.2047
Schalet, B.D., Pilkonis, P.A., Yu, L., Dodds, N., Johnston, K.L., Yount, S., . . . Cella, D. (2016). Clinical validity of PROMIS Depression, Anxiety, and Anger across diverse clinical samples. Journal of Clinical Epidemiology, 73, 119–127. https://doi.org/10.1016/j.jclinepi.2015.08.036
Teare, M.D., Dimairo, M., Shephard, N., Hayman, A., Whitehead, A., & Walters, S.J. (2014). Sample size requirements to estimate key design parameters from external pilot randomised controlled trials: A simulation study. Trials, 15, 264. https://doi.org/10.1186/1745-6215-15-264
Terwee, C.B., Peipert, J.D., Chapman, R., Lai, J.-S., Terluin, B., Cella, D., . . . Mokkink, L.B. (2021). Minimal important change (MIC): A conceptual clarification and systematic review of MIC estimates of PROMIS measures. Quality of Life Research, 30(10), 2729–2754. https://doi.org/10.1007/s11136-021-02925-y
Von Ah, D., Brown, C.G., Brown, S.J., Bryant, A.L., Davies, M., Dodd, M., . . . Cooley, M.E. (2019). Research Agenda of the Oncology Nursing Society: 2019–2022. Oncology Nursing Forum, 46(6), 654–669. https://doi.org/10.1188/19.ONF.654-669
Von Ah, D., & Crouch, A. (2020). Cognitive rehabilitation for cognitive dysfunction after cancer and cancer treatment: Implications for nursing practice. Seminars in Oncology Nursing, 36(1), 150977. https://doi.org/10.1016/j.soncn.2019.150977
Von Ah, D., Habermann, B., Carpenter, J.S., & Schneider, B.L. (2012). Impact of perceived cognitive impairment in breast cancer survivors. European Journal of Oncology Nursing, 17(2), 236–241. https://doi.org/10.1016/j.ejon.2012.06.002
Wefel, J.S., Lenzi, R., Theriault, R.L., Davis, R.N., & Meyers, C.A. (2004). The cognitive sequelae of standard-dose adjuvant chemotherapy in women with breast carcinoma: Results of a prospective, randomized longitudinal trial. Cancer, 100(11), 2292–2299. https://doi.org/10.1002/cncr.20272


