Neurotoxic Side Effects Early in the Oxaliplatin Treatment Period in Patients With Colorectal Cancer
Purpose: To identify and describe experiences of patients with colorectal cancer (CRC) who have neurotoxic side effects early in the oxaliplatin treatment period, and how neurotoxicity affects their daily lives.
Participants & Setting: 10 patients with stage II–III CRC were included. All were treated with adjuvant oxaliplatin postoperatively and assessed neurotoxicity via a platform-independent mobile phone–based system. Patients were recruited from two hospitals in southern Sweden from November 2013 to August 2014.
Methodologic Approach: Qualitative interview study conducted through open-ended, face-to-face, qualitative interviews. Thematic analysis was used.
Findings: A main theme was identified: “Endure neurotoxic side effects.” This theme illuminated how patients adapt to manage daily life. Patients’ daily lives, as well as their psychosocial well-being, were affected. Patients changed their daily routines and reprioritized activities.
Implications for Nursing: Nurses have an obligation to communicate the importance of early detection of neurotoxicity. Mobile phone technology seems to be a valuable tool for monitoring patient-reported neurotoxicity to improve communication and supportive care.
Jump to a section
Few studies have examined the experiences of patients with colorectal cancer (CRC) with oxaliplatin-induced neurotoxicity early in the chemotherapy treatment period and how these experiences affect their daily lives. Postoperative adjuvant chemotherapy consisting of 5-fluorouracil in combination with folinic acid (leucovorin) is an established treatment for patients with locally advanced CRC or with regional lymph node metastases. This regimen has relatively low toxicity, and most patients with cancer cope with it without troublesome side effects. By adding oxaliplatin, the relapse rate can be reduced by about 25%; however, the risk of side effects increases (Land et al., 2007). Oxaliplatin can cause acute and chronic neurotoxicity (e.g., cold sensitivity; numbness; tingling in the hands, feet, and face; pain) (Cavaletti & Marmiroli, 2015; Vatandoust et al., 2014). Another aspect of importance is that a correlation exists between the severities of acute and chronic neurotoxic side effects. Hyperacute neurotoxic side effects can be a predictor of oxaliplatin-induced persistent neurotoxicity (Tanishima et al., 2017).
Studies have shown that chronic neurotoxicity affects patients’ health-related quality of life (Bakitas, 2007; Tofthagen, Donovan, Morgan, Shibata, & Yeh, 2013; Tofthagen, McAllister, & McMillan, 2011). The number of people with CRC has increased, and more people with CRC are benefitting from adjuvant therapy (Seretny et al., 2014). The severity of neurotoxic side effects depends on the dose and duration of chemotherapy; consequently, the dosage of oxaliplatin is often reduced or treatment is stopped because of the fear of debilitating and prolonged neurotoxicity (de Gramont et al., 2007; Vatandoust et al., 2014).
To improve well-being during therapy, dose reduction could be useful in practice for patients who exhibit moderate or severe neurotoxicity during treatment. A cumulative dose of oxaliplatin is associated with chronic neurotoxicity, but even patients receiving dose reduction can be at risk for chronic neurotoxicity (Beijers et al., 2015). Previous studies also have shown that patients rate oxaliplatin-induced neurotoxicity more severely than healthcare providers do, and nurses lacked knowledge regarding neurotoxicity (Narita et al., 2015; Smith et al., 2014). Patients with cancer who are treated with chemotherapy struggle for survival and often experience different side effects and distress. Therefore, healthcare providers must be able to target information and support (Pedersen, Koktved, & Nielsen, 2013). To control side effects and distress, patients’ self-care abilities may be an important resource (Orem, Taylor, & Renpenning, 2001). Self-care can be influenced by many factors. The balance and relations between self-care and patients’ ability to care for themselves, both with and without support, can be a helpful consideration in nursing (Orem et al., 2001).
Because no treatment for oxaliplatin-induced neurotoxicity is generally accepted, with appropriate assessment, identifying these patients early is important to reduce the risk of their developing chronic neurotoxic side effects (Haryani, Fetzer, Wu, & Hsu, 2017; Padman et al., 2015). To the authors’ knowledge, no previous studies have examined patients with CRC experiencing neurotoxic side effects early in the treatment phase. The purpose of the current study is to identify and describe experiences of patients with CRC who have neurotoxic side effects early in the oxaliplatin treatment period, and how neurotoxicity affects their daily lives.
Methods
This qualitative study was conducted through open-ended, qualitative interviews and analyzed using thematic analysis (Braun & Clarke, 2006). The study was approved by the Regional Ethical Review Board in Linköping, Sweden (no. 2012/301-31), and patients with CRC were provided with oral and written informed consent in line with the Declaration of Helsinki.
Sample and Setting
The inclusion criteria were the following: being at least 18 years of age, having stage II–III CRC, being treated with adjuvant oxaliplatin postoperatively, having no history of neurotoxicity, and being able to read and speak Swedish and answer repeated questionnaires sent by mobile phone to detect neurotoxicity. Participants were included with purposeful and consecutive selection when they had reported neurotoxic side effects. Ten patients with CRC were included from November 2013 to August 2014. They were recruited from LinkÖping University Hospital and Länssjukhuset Ryhov, both in Sweden. Participants’ ages ranged from 44–68 years (median = 65 years, mean = 61 years). They were receiving oxaliplatin 175–520 mg/m2. Other characteristics of the participants are presented in Table 1. 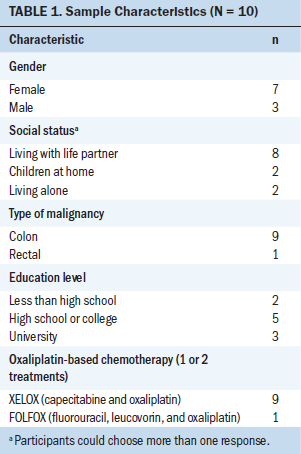
Procedure
Each individual’s neurotoxic side effects were self-reported via the oxaliplatin-associated neurotoxicity questionnaire (OANQ) using a mobile phone–based system, Circadian Questions (www.cqmobil.se), in real-time (Drott, Vilhelmsson, Kjellgren, & Berterö, 2016; Gustafsson, Litström, Berterö, & Drott, 2016; Leonard et al., 2005). The OANQ is divided into three sections corresponding to the side effect locations: upper extremities (10 items), lower extremities (9 items), and the face and mouth area (10 items). Participants first assessed whether they had the specific side effect (yes or no). If they answered yes, they graded the side effects on a five-point Likert-type scale ranging from 1 (hardly any) to 5 (very much). After that, they graded the impact on daily activities in a functional sense from 1 (hardly at all bothered) to 5 (extremely bothered) (Leonard et al., 2005). Tests of reliability (OANQ) were performed and reported. Internal consistency was strong for the three domains of the scale (alpha > 0.84). Test–retest indicated that the OANQ was stable. The weighted kappa for symptom items and daily activities items showed overall feasibility (Gustafsson et al., 2016). Each participant’s neurotoxic side effects were assessed at baseline before the initial oxaliplatin treatment, the day before oxaliplatin treatment, and two days after each oxaliplatin treatment (assessed every cycle).
The research team had the option to view the participants’ answers concerning the neurotoxic side effects as web-based graphs that could be viewed after logging into a web interface. When the professionals initiated the transfer of the questionnaire to the patient’s mobile phone, they used a calendar function to fill in the specific dates. The dates in the calendar function were set according to each individual’s chemotherapy regimen. The calendar was adapted to each specific patient, and the chosen questionnaire was sent out to the patient at the right moment to attain customized assessments. This calendar function was specifically developed for this project to enhance individual, customized measurements. The reported data were transferred to a secure database, and answers were available in real-time. Participants were contacted by the first author (JD) by telephone when neurotoxic side effects were assessed for the first time during treatment, and a face-to-face interview meeting was booked at an appropriate location. An interview guide was used to define areas to be explored but also to allow the interviewer and interviewee to diverge from the main topics (Patton, 2015). Each interview began with an introductory question inviting participants to talk about their neurotoxic side effects and how these affected their daily lives. Probing responses, such as, “Please tell more” or “Could you explain?” were used continuously during the interviews. The interviews were audio recorded and lasted 8–55 minutes (mean = 25 minutes). The eight-minute interview was conducted with a participant with a hearing loss impairment; the interview provided rich and interesting data to analyze. The interviews were transcribed verbatim by the first author.
Data Analysis
The verbatim transcribed interviews were processed; thematic analysis followed the phases according to the following (Braun & Clarke, 2006, 2013; Patton, 2015):
• Familiarizing with the data: Data were read and reread several times to enable the researchers to familiarize themselves with them. The transcripts were processed, and initial thoughts and ideas were noted.
• Generating initial codes: Codes were identified that involved features of the data that were considered pertinent to the research question and the aim. The codes were used to ensure the data were arranged in a coherent pattern and were considered in relation to the dataset as a whole. An initial thematic map is presented in Figure 1.
• Searching for themes
• Reviewing themes
• Defining and naming themes: These larger sections of data consisted of combinations of different codes, which could be similar or were considered to involve the same aspect of the data. All initial codes relevant to the research question were incorporated into a theme.
• Producing the report
To increase the trustworthiness of the findings, the analysis was subjected to peer-debriefing and was continuously scrutinized by the first author and an additional researcher (CB). Any discrepancies were resolved through discussion. To increase the trustworthiness of the findings, the analysis was peer-debriefed in the research team. Each phase of thematic analysis represents a reflective process over time, moving backward and forward between phases, which strengthens trustworthiness (Lincoln & Guba, 1985; Nowell, Norris, White, & Moules, 2017). [[{"fid":"47431","view_mode":"default","fields":{"format":"default","alignment":"","field_file_image_alt_text[und][0][value]":false,"field_file_image_title_text[und][0][value]":false},"link_text":null,"type":"media","field_deltas":{"1":{"format":"default","alignment":"","field_file_image_alt_text[und][0][value]":false,"field_file_image_title_text[und][0][value]":false}},"attributes":{"class":"media-element file-default","data-delta":"1"}}]]
Findings
The most common neurotoxic side effects were cold sensitivity and tingling in the hands, feet, and face. Daily life and social life were affected by these side effects. Participants did many things to manage neurotoxicity. One main theme was identified: “Endure neurotoxic side effects.”
All participants stated that they were living with a life-threatening disease and were also trying to manage the side effects of treatment. All of them endured the side effects while expecting cure. The most important thing in their minds at the beginning of treatment was survival; because of this, they were prepared to endure severe neurotoxic side effects. Cold sensitivity and tingling in the hands, feet, and face were the most common self-reported side effects. They endured the neurotoxicity and tried to manage their lives using different methods and adding a positive attitude, as illustrated by one participant who discussed thinking about the future: “My goal is to be free from cancer. I look forward to many good years in the future.”
Adaptation was needed to manage the neurotoxic side effects in daily life. Sometimes, adaptation was about allowing more time for a task. Retired participants said they could implement adaptation more, because others were not dependent on them and they had no schedule to follow. One participant said, “It is a great advantage that I do not have to feel stressed out, particularly about things I might have promised to do, but had to cancel. It is a great advantage.”
Participants with small children had caretaking responsibilities. Stressful situations occurred when the neurotoxicity side effects meant they were not able to help and support their children in different activities. According to one participant:
The children need to butter their sandwiches themselves, for example. Then the older brother helps his little brother to butter sandwiches, prepare milk, and so on. So, I just stand here and tell him what to do, and then he does it.
All participants had problems with coldness. To prevent tingling and painful neurotoxicity, they used different strategies. One strategy was to avoid triggers (i.e., avoid cold temperatures and cold things). This strategy affected their way of living and their social activities. Another strategy often used was keeping themselves and the things they used warm (i.e., drinking warm beverages, wearing more clothes, wearing socks and gloves, using warm water to reduce side effects in affected areas, and avoiding going outside on cold and windy days). Handling cold food and beverages (e.g., taking them out of the freezer or refrigerator) demands adaptation, such as using gloves. One participant shared, “This thing with the drinking has been tough. I cannot drink anything cold. It affects me a lot, because I cannot go out and drink what I want, or if I go away it is not possible.”
Participants had different strategies for handling the neurotoxic side effects affecting their daily lives. According to one participant:
It is very hard to drink something cold. I think that is the worst thing. I like to drink cold water. I try to drink cold liquids, because it not easy to get enough fluids. What should I do? I think this is really hard.
Some participants seemed to be more prepared if their healthcare providers had informed them beforehand about the oxaliplatin treatment. Other participants found it difficult to be prepared for neurotoxic side effects as well as other side effects. One participant said:
I received enough information and was very well informed before I got my treatment. It was both good and bad to know. I was prepared for side effects, but I was not prepared for such a quick and immediate effect.
One frequently used strategy was accepting help from family members, partners, and friends, who were used as assistants. If participants were supposed to carry on with activities as usual, they needed to plan these activities carefully. Another strategy used was changing routines they had for many years and creating new ones. One participant did the following:
I have eaten yogurt in the mornings for 40 years. I can do that now, and it is no problem, but I leave the yogurt at room temperature for maybe 20 minutes to half an hour before I sit down and eat it. Then I eat it slowly and carefully.
Changing habits and lifestyle affected participants emotionally. They had feelings of sadness at not being able to do everything as before. However, they adapted to the situation and had strategies for enduring these changes. Strategies for self-care to handle the neurotoxicity included receiving support and help from family members and avoiding activities. Participants reprioritized activities, used strategies to mitigate unpleasant feelings, and developed self-care strategies to restore normality.
Discussion
The findings add new knowledge about experiences of patients with CRC early in the treatment period. Participants in the current study experienced neurotoxic side effects after one or two cycles of chemotherapy. The participants had bothersome neurotoxicity despite short duration and low doses of oxaliplatin. The main findings showed that the participants endured the neurotoxic side effects while expecting cure. Enduring is the suppression of emotions, which makes a situation tolerable (Morse, 2001). The participants dealt with the side effects because the treatment was supposed to bring about a cure and restore health. Their strong desire to survive entailed adaptations and strategies to manage daily life despite the neurotoxic side effects. This is in agreement with the findings of a study by Chao, Wang, Hsu, and Wang (2015).
The willingness of people to endure side effects highlights the importance of early detection of neurotoxicity—being able to identify people who are at high risk for chronic neurotoxicity. Nurses have an obligation to communicate and support and, in dialogue, to help people with CRC make decisions and report early side effects. The participants’ neurotoxicity in the current study occurred in the initial phase, and increased exposure to oxaliplatin may induce chronic neurotoxicity later. The severity of neurotoxic side effects depends on the dose and duration of oxaliplatin (de Gramont et al., 2007; Vatandoust et al., 2014).
Another aspect of importance is that hyperacute neurotoxic side effects can be a predictor of oxaliplatin-induced persistent neurotoxicity (Tanishima et al., 2017). The participants perceived that the most important issue was survival, and because of that, they could endure severe neurotoxic side effects by adapting to them and using strategies to manage daily life. Actions performed were individual, depending on a participant’s life situation and circumstances. The participants had different experiences and used various ways to manage the situation.
A common strategy for handling neurotoxic side effects in daily life was avoiding things and situations that generated the neurotoxicity. The results showed that accepting support from family members or life partners was common. Family members and life partners were significant resources for the participants. A previous study by Sjovall, Gunnars, Olsson, and Thome (2011) pointed out that partners have an important role in helping patients with cancer achieve normality during the illness trajectory; the current findings are similar in this regard. A previous study of neurotoxicity showed that people with cancer had not expected neurotoxicity to affect their lives to the extent that it did (Bennett et al., 2012). In the current findings, these neurotoxic side effects, which at first glance seemed to be only physical and functional, also affected participants’ social lives and roles. The participants undertook self-care actions by adjusting their daily lives and minimizing the impact of neurotoxic side effects.
Retired participants and participants living alone seemed to adapt easier because of the possibility of taking their time and modifying situations, regardless of the people around or time schedules. Participants who lived alone also found living in the present easier; this finding is similar to that of an earlier study (Tan, Tien Tau, & Lai Meng, 2015). Participants with small children had problems coping because of their responsibility for the family. These results are in agreement with other studies pointing out that an individual’s personality and other stressors can influence adaptation and psychosocial needs (Husson, Vissers, Denollet, & Mols, 2015; Santin et al., 2015).
The current findings showed that positive thinking about the future was an important issue regarding survival. This is in agreement with other studies (Chen & Chang, 2012; Drageset, Lindstrøm, & Underlid, 2016). Survival is a strong driving force.
As the current findings show, the participants did not perceive their side effects to be severe, but they were distressed about the way the neurotoxicity barrier affected their ability to carry out daily activities. Participants changed daily life routines, reprioritized activities, and used strategies to mitigate the neurotoxicity. In other words, participants with CRC coped with their early neurotoxic side effects in different ways and developed self-care activities to restore normality.
Strength and Limitations
The main strength of the current study is the real-time study (mobile phone technology) without recollection bias. Another strength was the variety regarding ages and marital status. More women were included in the study because of the purposeful and consecutive sampling strategy. The gender distribution could have affected the findings. In terms of the occurrence of side effects, these gender differences were not acknowledged, and no mention was made of them in the literature. The sample size was small, as is common for a qualitative study. However, the findings from the data are sufficient to fulfill the goal. Aiming for trustworthiness of the findings, the analysis was peer-debriefed in the research team and a six-phased method was followed (Lincoln & Guba, 1985; Nowell et al., 2017). The six phases establishing trustworthiness during each phase of thematic analysis is a reflective process over time, moving back and forward between phases. The various steps in the research process make the research confirmable and provide an audit trail. More than one researcher was involved in the analysis during the initial stage and throughout the process, a procedure which is known as researcher triangulation.
Implications for Nursing
Self-care behaviors of patients with cancer can be influenced by a lot of environmental factors. Participants in the current study endured neurotoxic side effects by adapting to them and using strategies to manage daily life. Actions performed were individual, depending on a person’s life situation. The participants changed daily life routines, reprioritized activities, and used strategies to mitigate the neurotoxicity. They coped with their early neurotoxic side effects in different ways and developed self-care activities to restore normality.
A nurse’s responsibility is to support people with cancer in promoting self-care to maintain health. Nursing support in cancer care includes communication and active listening in the dialogue to help patients with cancer make decisions and report early side effects. Nursing support should be dynamic and may change over time. Individualized care, based on repeated real-time assessments during chemotherapy, could improve communication, self-care activities, and decision making. Mobile phone technology seems to be a valuable tool for monitoring self-reported neurotoxicity and improving supportive care. 
Conclusion
The findings of the current study add knowledge about how patients with CRC undergoing oxaliplatin chemotherapy endure neurotoxic side effects early in the treatment phase. Awareness of early neurotoxicity and its consequences is needed in clinical practice to ensure that proper actions are taken and support is provided.
The authors gratefully acknowledge all those who participated in this study.
About the Author(s)
Jenny Drott, RN, PhD, is a graduate of the Division of Nursing Science, Department of Medical and Health Sciences; Hans Starkhammar, MD, PhD, is a physician in the Department of Oncology; and Karin Kjellgren, RN, RNT, PhD, is a professor and Carina Berterö, RNT, MScN, PhD, is a professor and senior lecturer, both in the Division of Nursing Science, Department of Medical and Health Sciences, all at Linköping University in Sweden. This research was funded by grants from the Swedish Cancer Society and the Division of Nursing Science, Department of Medical and Health Sciences at Linköping University. Drott completed the data collection. All authors contributed to the conceptualization and design, provided the analysis, and contributed to the manuscript preparation. Drott can be reached at jenny.drott@liu.se, with copy to ONFEditor@ons.org. (Submitted March 2018. Accepted April 17, 2018.)
References
Bakitas, M.A. (2007). Background noise: The experience of chemotherapy-induced peripheral neuropathy. Nursing Research, 56, 323–331. https://doi.org/10.1097/01.NNR.0000289503.22414.79
Beijers, A.J., Mols, F., Tjan-Heijnen, V.C., Faber, C.G., van de Poll-Franse, L.V., & Vreugdenhil, G. (2015). Peripheral neuropathy in colorectal cancer survivors: The influence of oxaliplatin administration. Results from the population-based PROFILES registry. Acta Oncologica, 54, 463–469.
Bennett, B.K., Park, S.B., Lin, C.S., Friedlander, M.L., Kiernan, M.C., & Goldstein, D. (2012). Impact of oxaliplatin-induced neuropathy: A patient perspective. Supportive Care in Cancer, 20, 2959–2967. https://doi.org/10.1007/s00520-012-1428-5
Braun, V., & Clarke, V. (2006). Using thematic analysis in psychology. Qualitative Research in Psychology, 3, 77–101. https://doi.org/10.1191/1478088706qp063oa
Braun, V., & Clarke, V. (2013). Successful qualitative research: A practical guide for beginners. Los Angeles, CA: Sage.
Cavaletti, G., & Marmiroli, P. (2015). Chemotherapy-induced peripheral neurotoxicity. Current Opinion in Neurology, 28, 500–507. https://doi.org/10.1097/WCO.0000000000000234
Chao, Y.H., Wang, S.Y., Hsu, T.H., & Wang, K.W. (2015). The desire to survive: The adaptation process of adult cancer patients undergoing radiotherapy. Japan Journal of Nursing Science, 12, 79–86. https://doi.org/10.1111/jjns.12050
Chen, P.Y., & Chang, H.C. (2012). The coping process of patients with cancer. European Journal of Oncology Nursing, 16, 10–16. https://doi.org/10.1016/j.ejon.2011.01.002
de Gramont, A., Buyse, M., Abrahantes, J.C., Burzykowski, T., Quinaux, E., Cervantes, A., . . . Tournigand, C. (2007). Reintroduction of oxaliplatin is associated with improved survival in advanced colorectal cancer. Journal of Clinical Oncology, 25, 3224–3229. https://doi.org/10.1200/JCO.2006.10.4380
Drageset, S., Lindstrøm, T.C., & Underlid, K. (2016). “I just have to move on”: Women’s coping experiences and reflections following their first year after primary breast cancer surgery. European Journal of Oncology Nursing, 21, 205–211. https://doi.org/10.1016/j.ejon.2015.10.005
Drott, J., Vilhelmsson, M., Kjellgren, K., & Berterö, C. (2016). Experiences with a self-reported mobile phone-based system among patients with colorectal cancer: A qualitative study. JMIR mHealth and uHealth, 4(2), e66. https://doi.org/10.2196/mhealth.5426
Gustafsson, E., Litström, E., Berterö, C., & Drott, J. (2016). Reliability testing of oxaliplatin-associated neurotoxicity questionnaire (OANQ), a pilot study. Supportive Care in Cancer, 24, 747–754. https://doi.org/10.1007/s00520-015-2838-y
Haryani, H., Fetzer, S.J., Wu, C.L., & Hsu, Y.Y. (2017). Chemotherapy-induced peripheral neuropathy assessment tools: A systematic review [Online exclusive]. Oncology Nursing Forum, 44, E111–E123. https://doi.org/10.1188/17.ONF.E111–E123
Husson, O., Vissers, P.A., Denollet, J., & Mols, F. (2015). The role of personality in the course of health-related quality of life and disease-specific health status among colorectal cancer survivors: A prospective population-based study from the PROFILES registry. Acta Oncologica, 54, 669–677. https://doi.org/10.3109/0284186X.2014.996663
Land, S.R., Kopec, J.A., Cecchini, R.S., Ganz, P.A., Wieand, H.S., Colangelo, L.H., . . . Wolmark, N. (2007). Neurotoxicity from oxaliplatin combined with weekly bolus fluorouracil and leucovorin as surgical adjuvant chemotherapy for stage II and III colon cancer: NSABP C-07. Journal of Clinical Oncology, 25, 2205–2211. https://doi.org/10.1200/JCO.2006.08.6652
Leonard, G.D., Wright, M.A., Quinn, M.G., Fioravanti, S., Harold, N., Schuler, B., . . . Grem, J.L. (2005). Survey of oxaliplatin-associated neurotoxicity using an interview-based questionnaire in patients with metastatic colorectal cancer. BMC Cancer, 5, 116. https://doi.org/10.1186/1471-2407-5-116
Lincoln, Y.S., & Guba, E.G. (1985). Naturalistic inquiry. Thousand Oaks, CA: Sage.
Morse, J.M. (2001). Toward a praxis theory of suffering. Advances in Nursing Science, 24, 47–59. https://doi.org/10.1097/00012272-200109000-00007
Narita, Y., Taniguchi, H., Komori, K., Kimura, K., Kinoshita, T., Komori, A., . . . Muro, K. (2015). Differences in attitude toward adjuvant chemotherapy between colorectal cancer survivors and the medical staff of Japanese hospitals. International Journal of Clinical Oncology, 20, 755–760. https://doi.org/10.1007/s10147-014-0772-5
Nowell, L.S., Norris, J.M., White, D.E., & Moules, N.J. (2017). Thematic analysis: Striving to meet the trustworthiness criteria. International Journal of Qualitative Methods, 16, 1–13. https://doi.org/10.1177/1609406917733847
Orem, D.E., Taylor, S.G., & Renpenning, K.M. (2001). Nursing: Concepts of practice (6th ed.). St. Louis, MO: Mosby.
Padman, S., Lee, J., Kumar, R., Slee, M., Hakendorf, P., Richards, A., . . . Karapetis, C.S. (2015). Late effects of oxaliplatin-induced peripheral neuropathy (LEON)—Cross-sectional cohort study of patients with colorectal cancer surviving at least 2 years. Supportive Care in Cancer, 23, 861–869. https://doi.org/10.1007/s00520-014-2423-9
Patton, M.Q. (2015). Qualitative research and evaluation methods: Integrating theory and practice (5th ed.). Thousand Oaks, CA: Sage.
Pedersen, B., Koktved, D.P., & Nielsen, L.L. (2013). Living with side effects from cancer treatment—A challenge to target information. Scandinavian Journal of Caring Sciences, 27, 715–723. https://doi.org/10.1111/j.1471-6712.2012.01085.x
Santin, O., Murray, L., Prue, G., Gavin, A., Gormley, G., & Donnelly, M. (2015). Self-reported psychosocial needs and health-related quality of life of colorectal cancer survivors. European Journal of Oncology Nursing, 19, 336–342. https://doi.org/10.1016/j.ejon.2015.01.009
Seretny, M., Currie, G.L., Sena, E.S., Ramnarine, S., Grant, R., MacLeod, M.R., . . . Fallon, M. (2014). Incidence, prevalence, and predictors of chemotherapy-induced peripheral neuropathy: A systematic review and meta-analysis. Pain, 155, 2461–2470. https://doi.org/10.1016/j.pain.2014.09.020
Sjovall, K., Gunnars, B., Olsson, H., & Thome, B. (2011). Experiences of living with advanced colorectal cancer from two perspectives—Inside and outside. European Journal of Oncology Nursing, 15, 390–397. https://doi.org/10.1016/j.ejon.2010.11.004
Smith, E.M., Campbell, G., Tofthagen, C., Kottschade, L., Collins, M.L., Warton, C., . . . Visovsky, C. (2014). Nursing knowledge, practice patterns, and learning preferences regarding chemotherapy-induced peripheral neuropathy. Oncology Nursing Forum, 41, 669–679. https://doi.org/10.1188/14.ONF.669-679
Tan, P.Y., Tien Tau, L.C., & Lai Meng, O.Y. (2015). Living with cancer alone? The experiences of singles diagnosed with colorectal cancer. Journal of Psychosocial Oncology, 33, 354–376. https://doi.org/10.1080/07347332.2015.1045678
Tanishima, H., Tominaga, T., Kimura, M., Maeda, T., Shirai, Y., & Horiuchi, T. (2017). Hyperacute peripheral neuropathy is a predictor of oxaliplatin-induced persistent peripheral neuropathy. Supportive Care in Cancer, 25, 1383–1389. https://doi.org/10.1007/s00520-016-3514-6
Tofthagen, C., Donovan, K.A., Morgan, M.A., Shibata, D., & Yeh, Y. (2013). Oxaliplatin-induced peripheral neuropathy’s effects on health-related quality of life of colorectal cancer survivors. Supportive Care in Cancer, 21, 3307–3313. https://doi.org/10.1007/s00520-013-1905-5
Tofthagen, C., McAllister, R.D., & McMillan, S.C. (2011). Peripheral neuropathy in patients with colorectal cancer receiving oxaliplatin. Clinical Journal of Oncology Nursing, 15, 182–188. https://doi.org/10.1188/11.CJON.182-188
Vatandoust, S., Joshi, R., Pittman, K.B., Esterman, A., Broadbridge, V., Adams, J., . . . Price, T.J. (2014). A descriptive study of persistent oxaliplatin-induced peripheral neuropathy in patients with colorectal cancer. Supportive Care in Cancer, 22, 513–518. https://doi.org/10.1007/s00520-013-2004-3




