Impact of Diabetes on the Symptoms of Breast Cancer Survivors
Objectives: To examine the impact of diabetes on the symptoms of women with breast cancer.
Sample & Setting: 121 women with breast cancer who self-identified as having a diabetes diagnosis and 1,006 women with breast cancer without diabetes from 97 sites across the United States.
Methods & Variables: Symptom scores for depression, anxiety, sexual function, peripheral neuropathy, physical function, attention function, sleep disturbance, and fatigue were compared between women with breast cancer and diabetes and women with breast cancer without diabetes, controlling for age, education, income, marital status, and body mass index (BMI).
Results: Women with breast cancer and diabetes who were three to eight years postdiagnosis reported poorer physical and attention function, more sleep disturbance, and greater fatigue than women with breast cancer without diabetes. Age, education, income, and BMI were independent predictors of symptoms experienced by women with breast cancer.
Implications for Nursing: Oncology nurses can assess and monitor women with breast cancer and diabetes for increased post-treatment sequelae. If problematic symptoms are identified, implementing treatment plans can decrease symptom burden and increase quality of life for women with breast cancer and diabetes.
Jump to a section
According to the Centers for Disease Control and Prevention (2017), cancer and diabetes are leading causes of death in the United States. Breast cancer is the most common cancer diagnosed among women in the United States, with an estimated 268,600 new cases in 2019 (National Cancer Institute Surveillance, Epidemiology, and End Results Program, 2019). Women with diabetes have a 23% higher risk for developing breast cancer than women without diabetes (De Bruijn et al., 2013), and a preexisting diagnosis of diabetes is associated with a 37% risk for all-cause mortality among women with breast cancer (Shao et al, 2018; Zhou, Zhang, Gu, & Xia, 2015). In addition, the symptoms experienced by women with diabetes may be similar to those experienced by women with breast cancer, and diabetes can exacerbate breast cancer symptoms.
Because women with breast cancer are living longer, focusing on the role of comorbidities on long-term outcomes is important. Breast cancer and diabetes share common symptoms that affect quality of life (Fu et al., 2015), such as depression, anxiety, sexual dysfunction, decreased physical function, fatigue, and pain (Doong et al. 2015; Ferreira et al., 2014; Hershey, Given, Given, Von Eye, & You, 2012; Young-Hyman et al., 2016). Poor cognitive function, poor attention function (Ferreira et al., 2014; Von Ah, Habermann, Carpenter, & Schneider, 2013), and peripheral neuropathy are central nervous system complications that are also common to both diseases (Hershey, Given, et al., 2012; Tang et al., 2016; Vissers et al., 2014). Although these symptoms result from different etiologies in diabetes and breast cancer, they can be exacerbated when coexistence occurs.
Methods
Sample and Setting
Data from a cross-sectional quality-of-life study by Champion et al. (2014) were collected through the Eastern Cooperative Oncology Group–American College of Radiology Imaging Network (ECOG-ACRIN) Cancer Research Group. Results of the primary study are reported elsewhere (Champion et al., 2014). In the ECOG-ACRIN trial, breast cancer survivors received chemotherapy or combination chemotherapy and endocrine therapy regimens. Some women were also treated with surgical intervention, including mastectomy, lumpectomy, or radiation therapy. Eligibility for the primary study included being (a) diagnosed with breast cancer, (b) three to eight years post-treatment without recurrence at the time of recruitment, and (c) treated with a chemotherapy regimen of doxorubicin, paclitaxel, and cyclophosphamide. Women receiving surgical intervention or radiation therapy were also eligible. Additional criteria included residing in the United States, being aged 18 years or older, being able to speak and read English, and being able to complete a background questionnaire on their health history. Information on the study, including all study materials, was mailed to potential participants. Women were enrolled once they completed the background questionnaire and provided informed consent to participate.
Measures
Demographic variables and medical history were assessed through self-reported measures and confirmed by review of electronic health records. BMI was calculated from the self-reported height and weight measurements on participants’ questionnaires. The questionnaire assessed multiple aspects of quality of life, including physical, social, psychological, spiritual, and overall well-being. This study was approved by the Institutional Review Board (IRB) at Indiana University—Purdue University in Indianapolis and by IRBs for all 97 ECOG-ACRIN sites.
Sociodemographic variables, including age, race, income, education, marital status, self-reported BMI, and comorbidities, were collected. Participants were also asked to identify whether they had a diabetes diagnosis. Because of the retrospective nature of the study, the type of diabetes (type 1 or 2) was not captured.
Depression was measured using the Center for Epidemiologic Studies–Depression (CES-D) Scale (Radloff, 1977). This 20-item instrument measures the presence and severity of depressive symptoms. Validity and reliability of the CES-D have been reported as 0.9 (Cosco, Prina, Stubbs, & Wu, 2017; Radloff, 1977). Breast cancer survivors rated how often they experienced symptoms associated with depression on a scale of 0 to 3 (0 = rarely or none of the time, 1 = some or little of the time, 2 = moderately or much of the time, and 3 = most or almost all of the time) for each item. Scores ranged from 0 to 60, with higher scores indicating greater depressive symptoms. The Cronbach alpha for this sample was 0.89.
Anxiety was measured using the State–Trait Anxiety Inventory (Spielberger, Gorsuch, & Lushene, 1970). This 40-item scale measures the intensity of anxious feelings. Internal consistency and reliability have been reported as ranging from 0.86 to 0.95 (Julian, 2011; Spielberger, Gorsuch, Lushene, Vagg, & Jacobs, 1983). Higher scores indicate higher anxiety. The Cronbach alpha was 0.93.
The Index of Sexual Satisfaction is a 10-item scale that measures sexual interest, enjoyment, and function. A higher score indicates better sexual function (Hudson, Harrison, & Crosscup, 1981). Internal consistency and reliability have been reported as 0.89 (Santos Iglesias et al., 2009). The Cronbach alpha for this sample was 0.78.
The Symptom Survivor Checklist was used to assess peripheral neuropathy. This researcher-derived checklist is based on symptoms commonly associated with peripheral neuropathy. The scale includes three subsets of questions related to sensations on the side of the body where the women had cancer treatment. Higher scores indicate more symptoms. The Cronbach alpha coefficient for this sample was 0.78.
Physical functioning was measured using the 10-item Physical Functioning Scale (PF-10) (McHorney, Ware, Rogers, Raczek, & Lu, 1992). Internal consistency coefficients for this instrument were reported as ranging from 0.84 to 0.89 (Champion et al., 2014). The PF-10 is comprised of 10 items that ask participants to rank how their health limits their activities on a three-point scale of “yes, limited a lot” (1 point), “some limitations” (2 points), or “no, not limited at all” (3 points). Total scores ranged from 0 to 30, with higher scores indicating better physical functioning. The Cronbach alpha coefficient for this sample was 0.89.
The Attention Function Index is a 16-item scale that asks questions related to cognitive function during the past four weeks. Questions focus on planning and following through with daily activities or plans, as well as the time and effort that is involved (Cimprich, 1992, 1993). Internal consistency and reliability have been reported as 0.92 (Cimprich, Visovatti, & Ronis, 2011). Responses to each question range from 0 (lower capacity) to 10 (higher capacity), with higher total mean scores indicating better attention functioning. The Cronbach alpha coefficient for this sample was 0.933.
The Pittsburgh Sleep Quality Index (PSQI) is a 19-item self-report measure that asks survivors to rate seven aspects of sleep during a one-month interval. Each item is weighted on a 0 (not during the last month) to 3 (three or more times per week) scale. The global PSQI score is calculated by totaling the seven component scores and one composite score into an overall score ranging from 0 (healthy sleep quality) to 21 (poor sleep quality), with higher scores denoting poorer sleep quality (Buysse et al., 1991; Singh, Teel, Sabus, McGinis, & Kluding, 2016). Internal consistency and reliability have been reported as 0.83 (Smyth, 2012). A total score of 5 or greater indicates sleep disturbance (Zhu et al., 2018). The Cronbach alpha coefficient for this sample was 0.6.
The Functional Assessment of Cancer Therapy–Fatigue (FACT-F) is a 13-item scale that measures participants’ self-reported energy for daily activities in the past four weeks (Cella et al., 1993). An internal consistency of 0.95 has been previously reported (Yellen, Cella, Webster, Blendowski, & Kaplan, 1997). Responses to each question ranged from 0 (not at all) to 4 (very much so) and were reverse-scored so that higher scores indicated less fatigue. The Cronbach alpha coefficient for this sample was 0.94.
Data Analysis
Data analysis was conducted using IBM SPSS Statistics, version 24.0, and results were considered significant at p < 0.05. Normality of the data was confirmed in the primary study (Champion et al., 2014). Descriptive analyses of the means of the symptoms (depression, anxiety, sexual function, peripheral neuropathy, physical function, attention function, sleep disturbance, and fatigue) for women with breast cancer with and without a diabetes diagnosis were compared using t tests. Significant symptoms (physical function, attention function, sleep disturbance, and fatigue) were analyzed using linear regressions, which examined the relationship between diabetic status and potential covariates (age, BMI, education, income, and marital status) on each of the dependent variables.
Results
Descriptive Statistics
The dataset for this analysis included women with breast cancer (N = 1,127), 11% (n = 121) of whom self-reported a comorbid diabetes diagnosis. Women with breast cancer were primarily Caucasian, married, middle-aged, and well educated, with an income level of less than $75,000. Sample characteristics are listed in Table 1. Variables that were significantly different between diabetics and nondiabetics were used as covariates. 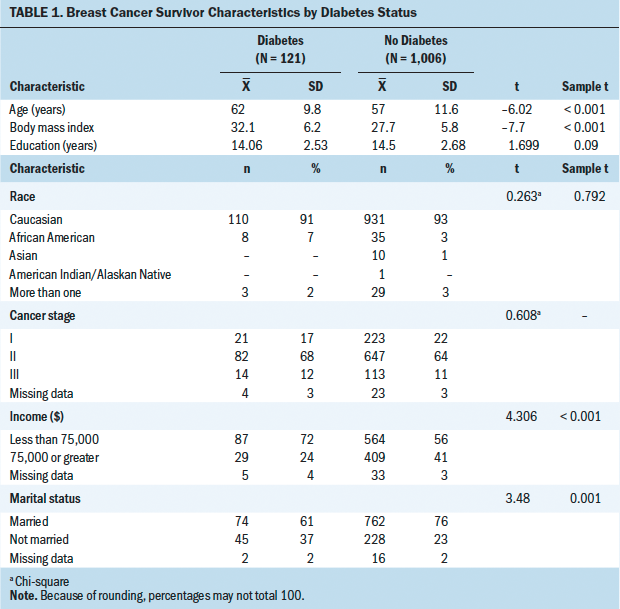
Independent sample t tests indicated significantly different mean scores among women with breast cancer and diabetes for four of the eight symptoms. Women with breast cancer and diabetes reported poorer physical function (p < 0.001), lower attention function (p = 0.008), greater sleep disturbance (p = 0.019), and greater fatigue (p = 0.013) compared to breast cancer survivors without diabetes. No significant differences among depression, anxiety, sexual function, and peripheral neuropathy were indicated. Mean symptom scores are listed in Table 2. Physical function, attention function, sleep disturbance, and fatigue were regressed on diabetic status while controlling for significantly different variables between diabetics and nondiabetics, including age, BMI, marital status, and income (see Table 3). 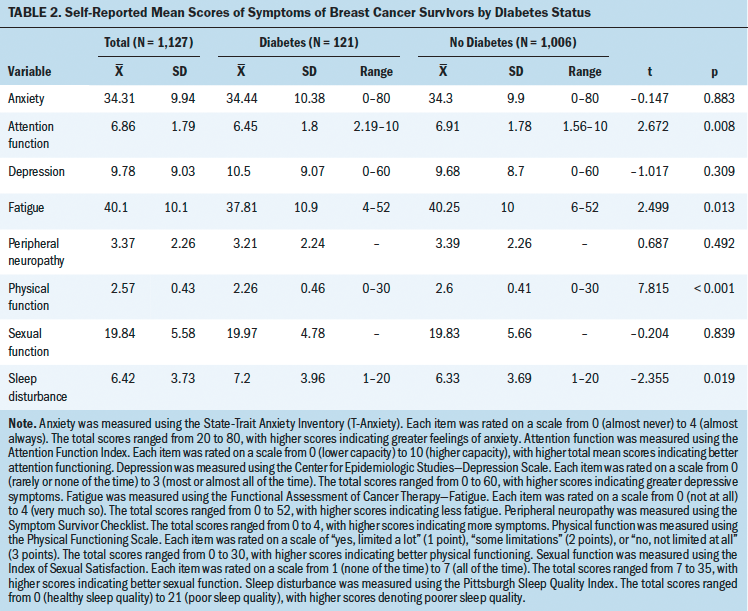
Women with breast cancer and diabetes had poorer physical function than those without diabetes (p < 0.001). As age increased, physical function decreased (p < 0.001). Better education (p < 0.001) and greater income (p < 0.001) were positively related to physical function. Marital status was not a significant predictor of physical function (p = 0.97). Women with breast cancer who were obese compared to those of normal weight reported poorer physical function (p < 0.001). [[{"fid":"51246","view_mode":"default","fields":{"format":"default","alignment":"","field_file_image_alt_text[und][0][value]":false,"field_file_image_title_text[und][0][value]":false},"link_text":null,"type":"media","field_deltas":{"1":{"format":"default","alignment":"","field_file_image_alt_text[und][0][value]":false,"field_file_image_title_text[und][0][value]":false}},"attributes":{"class":"media-element file-default","data-delta":"1"}}]]
Attention function scores were significantly lower in women with breast cancer and diabetes compared to those without diabetes (p = 0.003). Age (p < 0.001), education (p < 0.001), and income (p = 0.004) were positively related to attention function, indicating that women with breast cancer with more years of education and higher income had greater attention function. Marital status was not a significant predictor (p = 0.547). Attention function was significantly lower in women with breast cancer who were overweight (p = 0.024) or obese (p = 0.043).
In this sample, women with breast cancer and diabetes reported experiencing more sleep disturbance than those without diabetes (p = 0.026). Age (p < 0.001), education (p = 0.003), and income (p = 0.006) were negatively related to sleep disturbance. Marital status did not predict sleep disturbance (p = 0.616). BMI was not a significant predictor of sleep disturbance among women with breast cancer.
Women with breast cancer and diabetes reported significantly greater fatigue (p = 0.033) than those without diabetes. Those who were older (p < 0.001), with more years of education (p = 0.042), and with higher income (p < 0.001) reported less fatigue. Marital status was not significant (p = 0.593). BMI predicted fatigue among the women with breast cancer who were overweight (p = 0.042) or obese (p < 0.001).
Because women with breast cancer and diabetes had a significantly higher BMI than those without diabetes the interaction between diabetes and BMI was tested as an additional variable in predicting each of the four symptoms. No significant interaction effects between BMI status (overweight or obese) and diabetes on the symptoms were determined.
Discussion
Although some studies found the prevalence of diabetes among women with breast cancer to be as high as 33% (Srokowski, Fang, Hortobagyi, & Giordano, 2009), the prevalence of breast cancer survivors with diabetes (11%) in this study is similar to other researchers who found that 12% of breast cancer survivors reported a diagnosis of diabetes (Fu et al., 2015; Tang et al., 2016). The results of this study indicated that the presence of diabetes in women with breast cancer was associated with poorer physical and attention function and greater sleep disturbance and fatigue.
In this study, the presence of diabetes negatively influenced physical function. These results are consistent with researchers who noted that women with breast cancer with comorbid diabetes reported significantly poorer physical function than women without diabetes following active treatment (Tang et al., 2016). In a study by Hershey, Tipton, Given, and Davis (2012), women with breast cancer and diabetes reported poorer physical function than those without diabetes. However, physical function was measured during treatment with chemotherapy, which may have influenced the results (Hershey, Tipton, et al., 2012). Chemotherapy treatment regimens may exacerbate symptoms in women with breast cancer and diabetes. In addition, the acute side effects of treatment were shown to reduce diabetes self-management activities (Hershey, Tipton, et al., 2012). Additional research is needed to determine whether physical function is more severely affected in women with breast cancer and diabetes.
As age increased, women with breast cancer reported poorer physical function. Increasing age is accompanied by an overall decline in physical function, affecting the ability to complete activities of daily living (Brady & Straight, 2014). Women with breast cancer who had higher education and income reported higher physical function. Higher education and income have been associated with an increased ability to engage in physical activity and healthy living activities, as well as better access to health care, which aids in sustaining physical function (Chetty et al., 2016). As expected, increased BMI (obesity only) was associated with poorer physical function. Higher amounts of body fat were associated with poorer physical performance, as well as greater limitations in functional ability and disability among community-dwelling older women (Rolland et al., 2009). Increased adiposity and obesity often occur concurrently with aging and are contributing factors to decreased muscle mass, capacity, and strength, which can result in poorer physical function (Brady & Straight, 2014; Schaap, Koster & Visser, 2013).
Although insignificant, women with breast cancer and diabetes reported fewer symptoms of peripheral neuropathy than women with breast cancer. Because people with diabetes can present with baseline peripheral sensory impairment, making them less sensitive to additional sensory loss, patients without diabetes are more likely to recognize and be affected by sensory changes. Additional studies examining peripheral neuropathy in women with breast cancer and without diabetes are warranted.
This is the first study to examine attention function in the context of women with breast cancer and diabetes. Women with breast cancer and diabetes reported poorer attention function than their counterparts without diabetes. Women with breast cancer frequently report changes in cognitive function and attention as a troublesome symptom (Frank, Vance, Jukkala, & Meneses, 2014; Von Ah et al., 2009; Von Ah, Storey, Crouch, et al. 2016; Von Ah, Storey, Tallman, et al. 2016), which can persist for as many as 20 years after completing treatment for breast cancer (Koppelmans et al., 2012). Cancer treatment and hormonal changes are contributing factors to poorer attention function among women with breast cancer. Independent of a cancer diagnosis, patients with diabetes also reported decreased cognitive function (Feinkohl, Price, Strachan, & Frier, 2015; Koekkoek, Kappelle, van den Berg, Rutten, & Biessels, 2015; Mansur et al., 2014; Zilliox, Chadreasekaran, Kwan, & Russell, 2016), particularly in the domains of memory, processing speed, executive function (Koekkoek, et al., 2015), and attention function (Monette, Baird, & Jackson, 2014; Zilliox et al., 2016). In a meta-analysis of 25 studies on people with diabetes, researchers found statistically significantly poorer cognitive function (including attention function) among those with diabetes (p < 0.05) (Monette et al., 2014). Lack of glycemic control (hypoglycemia or hyperglycemia) suggests a contributing factor to these cognitive symptoms (Feinkohl et al., 2015; Seetharaman et al., 2015). Future studies can help to identify women with breast cancer who may be at a higher risk for impaired attention function. A better understanding of the impact of diabetes in women with breast cancer and its influence on attention function is important so that baseline and ongoing cognitive assessments can be conducted throughout the cancer trajectory.
Although previous research has demonstrated that increasing age is associated with poorer attention function, this study found that age was positively associated with attention function. In particular, younger women with breast cancer perceived poorer attention function than older women with breast cancer. This finding was also noted in the primary study that consisted of an equal sample of younger and older women with breast cancer. Champion et al. (2014) suggest that younger women with breast cancer are engaged in activities that require higher cognitive demands than older women with breast cancer; therefore, their perception of attention function is based on their higher demands. In a study by Von Ah et al. (2013) attention function in younger women with breast cancer was worse than it was in older women.
Similar to the findings of this study, higher education and income was associated with better cognition and attention function throughout the aging continuum (Lyu & Burr, 2016). In addition, researchers have noted the negative effect of higher BMI on cognitive abilities (Gunstad et al., 2007).
Women with breast cancer and diabetes reported more sleep disturbance than those without diabetes. This is the second report examining the impact of diabetes on sleep disturbance in women with breast cancer (Tang et al., 2016). Among women with breast cancer, sleep disturbance is reported with symptom onset from initial diagnosis throughout the cancer trajectory (Ancoli-Israel et al., 2014; Otte, Carpenter, Russell, Bigatti, & Champion, 2010). According to a study by Otte et al. (2010), women with breast cancer scored higher on the PSQI, indicating poorer sleep quality and greater sleep disturbance than women without breast cancer (p < 0.05). Similarly, in a longitudinal study by Ancoli-Israel et al. (2014), women with breast cancer had poorer sleep quality and more sleep disturbance than the noncancer controls (p < 0.05). Patients with diabetes reported poorer sleep quality and more sleep disturbance than people without diabetes (Barnard et al., 2016; Reutrakul et al., 2016; Zhu et al., 2018). Sleep disturbance in people with diabetes is linked to glucose metabolism (Spiegel, Tasali, Leproult, & Van Cauter, 2009) and subsequent suboptimal glycemic control (Reutrakul et al., 2016). Alterations in blood glucose can result in hypoglycemia, polydipsia, and polyuria, which can contribute to interrupted sleep patterns. Additional research is needed to examine the role of diabetes on the sleep patterns of women with breast cancer because sleep disturbance may exacerbate the severity of other symptoms, such as fatigue and depression.
In this study, younger women with breast cancer reported more sleep disturbance than older women with breast cancer. This is consistent with findings from the primary study that noted that younger women with breast cancer reported more sleep disturbance than older women with breast cancer (Champion et al., 2014). Similarly, in a study of 492 younger women with breast cancer (mean age = 48 years), researchers found that sleep disturbance was associated with postmenopausal symptoms, poorer physical function, and comorbid conditions (Otte et al., 2010). As education and income increased, sleep disturbance decreased. Similar findings were noted by researchers who found that those with higher socioeconomic status reported less sleep disturbances than those with lower socioeconomic status (Grandner et al., 2010; Mezick et al., 2008). In the current study, BMI was not associated with sleep disturbance. Although this finding differs from other research that links BMI to poorer sleep, this study used the PSQI to measure sleep disturbance specifically, whereas the others measured the duration of sleep, rapid eye movement sleep, obstructive sleep apnea, and daytime sleepiness in relation to BMI (Drager, Togeiro, Polotsky, & Lorenzi-Filho, 2013; Ford et al., 2014; Moraes et al., 2013).
Women with breast cancer and diabetes reported greater fatigue than those without diabetes. Only one existing study assessed fatigue in women with breast cancer with and without diabetes (Tang et al., 2016). Using the European Organisation for Research and Treatment of Cancer Quality of Life Questionnaire-Core 30, researchers noted higher fatigue scores (p < 0.05) among women with breast cancer and diabetes than in women with breast cancer only (Tang et al., 2016). Fatigue is a multidimensional symptom experienced by women with breast cancer and patients diagnosed with diabetes. Similar factors that contribute to fatigue were identified among women with breast cancer and diabetes, including poor physical function, poor sleep, cognitive dysfunction, anxiety, and depression (Champion et al., 2014; Otte et al., 2010; Park, Park, Quinn, & Fritschi, 2015; Singh et al., 2016). In addition, the physiologic demands of cancer, cancer treatment, and diabetes may decrease energy production in the body and worsen fatigue.
In this study, age was inversely associated with fatigue (p < 0.001). This is congruous with the primary study, which found that younger women with breast cancer reported more fatigue than older women (Champion et al., 2014). In a study by Garabeli Cavalli Klutcovsky et al. (2012) of 202 women with breast cancer (stage III) who were two to five years post-treatment, younger women reported higher levels of fatigue than older women. Similar to other studies, education was inversely related to fatigue (p = 0.037). Being overweight (p = 0.041) or obese (p < 0.001) also increased fatigue levels. Higher levels of fatigue and decreased physical function and activity have been found in women with breast cancer with higher BMI (Herath, Peswani, & Chitambar, 2016; Sheng, Sharma, Jerome, & Santa-Maria, 2019). In a study of primarily urban African American women, those with a higher BMI (30 kg/m2 or greater) reported more severe fatigue than women with a normal BMI (Jarosz et al., 2014). Because women with diabetes tend to have a higher BMI, women with breast cancer and a diagnosis of diabetes are at an increased risk for fatigue.
Strengths and Limitations
This study was the first to assess the impact of diabetes on eight symptoms commonly experienced by breast cancer survivors and patients with diabetes. The strength of the study was a robust sample size, with similar disease staging and treatment for breast cancer. This study relied on a self-report of diabetes, which limited the ability to determine whether the women were diagnosed with type 1 or 2 diabetes. Different pathologies are associated with each type, which may influence the symptoms experienced and their severity. The diabetes status of the women with breast cancer may have been underreported because estimates indicate that 7.2 million Americans do not know they have the condition (Centers for Disease Control and Prevention, 2017). Because of the retrospective study design, whether blood glucose levels influenced the type and severity of symptoms reported was not assessed. Because of the lack of diversity in this study, the findings cannot be generalized to women with breast cancer with diabetes of other ethnicities. Additional prospective studies with larger sample sizes are needed to confirm these findings and to identify the most prevalent symptoms experienced by women with breast cancer and diabetes. These findings can lead to the development of self-management strategies and interventions to mitigate symptoms and improve quality of life for women with breast cancer and diabetes.
Implications for Nursing
This study provides preliminary evidence that women with breast cancer and diabetes experience a greater symptom profile than women with breast cancer without diabetes. As women with breast cancer are living longer with comorbid conditions, it is important for oncology nurses to educate themselves on diabetes and breast cancer, as well as the potential effects that coexisting conditions can have on symptoms during the cancer trajectory. Oncology nurses can assess and identify women with breast cancer and diabetes as being at a higher risk for poor post-treatment sequelae. Diabetes status in women with breast cancer should be considered a risk factor for increased symptoms following treatment, and patients with diabetes should be closely monitored for symptoms. Baseline assessments of these symptoms at diagnosis and prior to the initiation of the treatment regimen, as well as ongoing assessment throughout the survivorship trajectory, are important to determine the unique impact of diabetes. 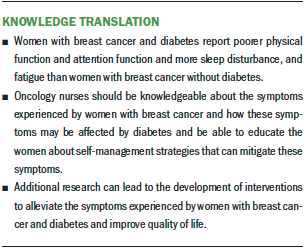
Conclusion
This study reported the effect of diabetes on four prominent symptoms experienced by breast cancer survivors who were three to eight years post-treatment and in remission. According to the findings in this study, women with breast cancer and diabetes are vulnerable to a greater symptom profile during the cancer trajectory than women without diabetes. Additional research on the impact of diabetes on symptoms experienced by women with breast cancer can help to develop and implement tailored interventions that can mitigate these symptoms. 
About the Author(s)
Susan Storey, PhD, RN, AOCNS®, and Andrea Cohee, PhD, RN, are assistant professors, and Wambui G. Gathirua-Mwangi, PhD, MPH, and Eric Vachon, PhD, RN, are postdoctoral fellows, all in the School of Nursing, Patrick Monahan, PhD, is a professor in the School of Medicine and the Richard M. Fairbanks School of Public Health, Julie Otte, PhD, RN, OCN®, is an associate professor in the School of Nursing, and Timothy E. Stump, MA, is a biostatistician in the School of Medicine, all at Indiana University in Indianapolis; David Cella, PhD, is chair of the Department of Medical Social Sciences at Northwestern University in Evanston, IL; and Victoria Champion, PhD, RN, FAAN, is a distinguished professor in the School of Nursing at Indiana University in Indianapolis. This study was coordinated by the Eastern Cooperative Oncology Group–American College of Radiology Imaging Network Cancer Research Group (Peter J. O’Dwyer, MD, and Mitchell D. Schnall, MD, PhD, group co-chairs) and funded by grants (CA189828, CA180795) from the National Cancer Institute of the National Institutes of Health. Mention of specific products and opinions related to those products do not indicate or imply endorsement by the Oncology Nursing Society. Storey, Gathirua-Mwangi, Cella, and Champion contributed to the conceptualization and design. Storey, Cohee, and Champion completed the data collection. Storey, Cohee, Monahan, Stump, and Champion provided statistical support. Storey, Gathirua-Mwangi, Vachon, Monahan, Otte, Stump, and Champion provided the analysis. All authors contributed to the manuscript preparation. Storey can be reached at sustorey@iu.edu, with copy to ONFEditor@ons.org. (Submitted November 2018. Accepted January 28, 2019.)
References
Ancoli-Israel, S., Liu., L., Rissling, M., Natarajan, L., Neikrug, A.B., Palmer, B.W., . . . Maglione, J. (2014). Sleep, fatigue, depression, and circadian activity rhythms in women with breast cancer before and after treatment: A 1-year longitudinal study. Supportive Care in Cancer, 22, 2535–2545. https://doi.org/10.1007/s00520-014-2204-5
Barnard, K., James, J., Kerr, D., Adolfsson, P., Runion, A., & Serbedzija, G. (2016). Impact of chronic sleep disturbance for people living with T1 diabetes. Journal of Diabetes Science and Technology, 10, 762–767. https://doi.org/10.1177/1932296815619181
Brady, A.O., & Straight, C.R. (2014). Muscle capacity and physical function in older women: What are the impacts of resistance training? Journal of Sport and Health Science, 3, 179–188. https://doi.org/10.1016/j.jshs.2014.04.002
Buysse, D.J., Reynolds, C.F., 3rd, Monk, T.H., Hoch, C.C., Yeager, A.L., & Kupfer, D.J. (1991). Quantification of subjective sleep quality in healthy elderly men and women using the Pittsburgh Sleep Quality Index (PSQI). Sleep, 14, 331–38.
Cella, D.F., Tulsky, D.S., Gray, G., Sarafian, B., Linn, E., Bonomi, A., . . . Brannon, J. (1993). The Functional Assessment of Cancer Therapy scale: Development and validation of the general measure. Journal of Clinical Oncology, 11, 570–579.
Centers for Disease Control and Prevention. (2017). National cen-ter for health statistics: Deaths and mortality. Retrieved from https://www.cdc.gov/nchs/fastats/deaths.htm
Champion, V.L., Wagner, L.I., Monahan, P.O., Daggy, J., Smith, L., Cohee, A, . . . Sledge, G.W., Jr. (2014). Comparison of younger and older breast cancer survivors and age-matched controls on specific and overall quality of life domains. Cancer, 120, 2237–2246. https://doi.org/10.1002/cncr.28737
Chetty, R., Stepner, M., Abraham, S., Lin, S., Scuderi, B., Turner, N., . . . Cutler, D. (2016). The association between income and life expectancy in the United States, 2001-2014. JAMA, 316, 1750–1766. https://doi.org/10.1001/jama.2016.4226.
Cimprich, B. (1992). Attentional fatigue following breast cancer surgery. Research in Nursing and Health, 15, 199–207.
Cimprich, B. (1993). Development of an intervention to restore attention in cancer patients. Cancer Nursing, 16, 83–92.
Cimprich, B., Visovatti, M., & Ronis, D.L. (2011). The Attention Function Index—A self-report cognitive measure. Psycho-Oncology, 20, 194–202. https://doi.org/10.1002/pon.1729
Cosco, T.D., Prina, M., Stubbs, B., & Wu, Y.T. (2017). Reliability and validity of the Center for Epidemiologic Studies Depression Scale in a population-based cohort of middle-aged U.S. adults. Journal of Nursing Measurement, 25, 476–485. https://doi.org/10.1891/1061-3749.25.3.476
De Bruijn, K.M., Arends, L.R., Hansen, B.E., Leeflang, S., Ruiter, R., & van Eijck, C.H. (2013). Systematic review and meta-analysis of the association between diabetes mellitus and incidence and mortality in breast and colorectal cancer. British Journal of Surgery, 100, 1421–1429. https://doi.org/10.1002/bjs.9229
Doong, S.H., Dhruva, A., Dunn, L.B., West, C., Paul, S.M., Cooper, B.A., . . . Miaskowski, C. (2015). Associations between cyto-kine genes and a symptom cluster of pain, fatigue, sleep disturbance, and depression in patients prior to breast cancer surgery. Biological Research for Nursing, 17, 237–247. https://doi.org/10.1177/1099800414550394
Drager, L.F., Togeiro, S.M., Polotsky, V.Y., & Lorenzi-Filho, G. (2013). Obstructive sleep apnea: A cardiometabolic risk in obesity and the metabolic syndrome. Journal of the American College of Cardiology, 62, 569-576. https://doi.org/10.1016/j.jacc.2013.05.045
Feinkohl, I., Price, J.F., Strachan, M.W., & Frier, B.M. (2015). The impact of diabetes on cognitive decline: Potential vascular, metabolic, and psychosocial risk factors. Alzheimer’s Research and Therapy, 7, 46. https://doi.org/10.1186/s13195-015-0130-5
Ferreira, M.C., Tozatti, J., Fachin, S.M., Oliveira, P.P., Santos, R.F., & Silva, M.E. (2014). Reduction of functional mobility and cognitive capacity in type 2 diabetes mellitus. Arquivos Brasileiros de Endocrinologia e Metabologia, 58, 946–952. https://doi.org/10.1590/0004-2730000003097
Ford, E.S., Li, C., Wheaton, A.G., Chapman, D.P., Perry, G.S., & Croft, J.B. (2014). Sleep duration and body mass index and waist circumference among U.S. adults. Obesity, 22, 598–607. https://doi.org/10.1002/oby.20558
Frank, J.S., Vance, D.E., Jukkala, A., & Meneses, K.M. (2014). Attention and memory deficits in breast cancer survivors: Implications for nursing practice and research. Journal of Neuroscience Nursing, 46, 274–284. https://doi.org/10.1097/JNN.0000000000000078
Fu, M.R., Axelrod, D., Guth, A.A., Cleland, C.M., Ryan, C.E., Weaver, K.R., . . . Melkus, G.D. (2015). Comorbidities and quality of life among breast cancer survivors: A prospective study. Journal of Personalized Medicine, 5, 229–242. https://doi.org/10.3390/jpm5030229
Garabeli Cavalli Kluthcovsky, A.C., Urbanetz, A.A., de Carvalho, D.S., Pereira Maluf, E.M., Schlickmann Sylvestre, G.C., & Bonatto Hatschbach, S.B. (2012). Fatigue after treatment in breast cancer survivors: Prevalence, determinants and impact on health-related quality of life. Supportive Care in Cancer, 20, 1901–1909. https://doi.org/10.1007/s00520-011-1293-7
Grandner, M.A., Patel, N.P., Gehrman, P.R., Xie, D., Sha, D., Weaver, T., & Gooneratne, N. (2010). Who gets the best sleep? Ethnic and socioeconomic factors related to sleep complaints. Sleep Medicine, 11, 470–478. https://doi.org/10.1016/j.sleep.2009.10.006
Gunstad, J., Paul, R.H., Cohen, R.A., Tate, D.F., Spitznagel, M.B., & Gordon, E. (2007). Elevated body mass index is associated with executive dysfunction in otherwise healthy adults. Comprehensive Psychiatry, 48, 57–61. https://doi.org/10.1016/j.comppsych.2006.05.001
Herath, K., Peswani, N., & Chitambar, C.R. (2016). Impact of obe-sity and exercise on chemotherapy-related fatigue. Supportive Care in Cancer, 24, 4257–4262. https://doi.org/10.1007/s00520-016-3257-4
Hershey, D.S., Given, B., Given, C., Von Eye, A., & You, M. (2012). Diabetes and cancer: Impact on health-related quality of life. Oncology Nursing Forum, 39, 449–457. https://doi.org/10.1188/12.ONF.449-457
Hershey, D.S., Tipton, J., Given, B., & Davis, E. (2012). Perceived impact of cancer treatment on diabetes self-management. Diabetes Educator, 38, 779–790.
Hudson, W.W., Harrison, D.F., & Crosscup, P.C. (1981). A short-form scale to measure sexual discord in dyadic relationships. Journal of Sex Research, 17, 157–174.
Jarosz, P.A., Davis, J.E., Yarandi, H.N., Farkas, R., Feingold, E., Shippings, S.H., . . . Williams, D. (2014). Obesity in urban women: Associations with sleep and sleepiness, fatigue and activity. Women’s Health Issues, 24, e447–e454. https://doi.org/10.1016/j.whi.2014.04.005
Julian, L.J. (2011). Measures of anxiety: State-Trait Anxiety Inventory (STAI), Beck Anxiety Inventory (BAI), and Hospital Anxiety and Depression Scale-Anxiety (HADS-A). Arthritis Care and Research, 63(Suppl. 11), S467–S472. https://doi.org/10.1002/acr.20561
Koekkoek, P.S., Kappelle, L.J., van den Berg, E., Rutten, G.E., & Biessels, G.J. (2015). Cognitive function in patients with diabetes mellitus: Guidance for daily care. Lancet Neurology, 14, 329–340. https://doi.org/10.1016/S1474-4422(14)70249-2
Koppelmans, V., Breteler, M.M., Boogerd, W., Seynaeve, C., Gundy, C., & Schagen S.B. (2012). Neuropsychological performance in survivors of breast cancer more than 20 years after adjuvant chemotherapy. Journal of Clinical Oncology, 30, 1080–1086. https://doi.org/10.1200/JCO.2011.37.0189
Lyu, J., & Burr, J.A. (2016). Socioeconomic status across the life course and cognitive function among older adults: An examination of the Latency, Pathways, and Accumulation Hypotheses. Journal of Aging and Health, 28, 40–67. https://doi.org/10.1177/0898264315585504
Mansur, R.B., Cha, D.S., Woldeyohannes, H.O., Scozynska, J.K., Zugman, A., Brietzke, E., & McIntyre, R.S. (2014). Diabetes mellitus and disturbances in brain connectivity: A bidirectional relationship? NeuroMolecular Medicine, 16, 658–668. http://doi.org/10.1007/s12017-014-8316-8
McHorney, C.A., Ware, J.E. Jr., Rogers, W., Raczek, A.E., & Lu, J.F. (1992). The validity and relative precision of MOS short- and long-form health status scales and Dartmouth COOP charts. Results from the Medical Outcomes Study. Medical Care, 30(5 Suppl.), MS253–MS265.
Mezick, E.J., Matthews, K.A., Hall, M., Strollo, P.J. Jr., Buysse, D.J., Kamarck, T.W., . . . Reis, S.E. (2008). Influence of race and socioeconomic status on sleep: Pittsburgh sleep SCORE project. Psychosomatic Medicine, 70, 410–416. https://doi.org/10.1097/PSY.0b013e31816fdf21
Monette, M.C., Baird, A., & Jackson, D.L. (2014). A meta-analysis of cognitive functioning in nondemented adults with type 2 diabetes mellitus. Canadian Journal of Diabetes, 38, 401–408. https://doi.org/10.1016/j.jcjd.2014.01.014
Moraes, W., Poyares, D., Zalcman, I., de Mello, M.T., Bittencourt, L.R., Santos-Silva, R., & Tufik, S. (2013). Association between body mass index and sleep duration assessed by objective methods in a representative sample of the adult population. Sleep Medicine, 14, 312–318. https://doi.org/10.1016/j.sleep.2012.11.010
National Cancer Institute Surveillance, Epidemiology, and End Results Program. (2019). Cancer stat facts: Female breast cancer. Retrieved from https://seer.cancer.gov/statfacts/html/breast.html
Otte, J.L., Carpenter, J.S., Russell, K.M., Bigatti, S., & Champion, V.L. (2010). Prevalence, severity, and correlates of sleep-wake disturbances in long-term breast cancer survivors. Journal of Pain and Symptom Management, 39, 535-547. https://doi.org/10.1016/j.jpainsymman.2009.07.004
Park, H., Park, C., Quinn, L., & Fritschi, C. (2015). Glucose con-trol and fatigue in type 2 diabetes: The mediating roles of diabetes symptoms and distress. Journal of Advanced Nursing, 71, 1650–1660. https://doi/org/10.1111/jan.12632
Radloff, L.S. (1977). The CES-D scale: A self-report depression scale for research in the general population. Applied Psychological Measurement, 1, 385–401. https://doi.org/10.1177/014662167700100306
Reutrakul, S., Thakkinstian, A., Anothaisintawee, T., Chontong, S., Borel, A.L., Perfect, M.M., . . . Knutson, K.L. (2016). Sleep characteristics in type 1 diabetes and associations with glycemic control: Systematic review and meta-analysis. Sleep Medicine, 23, 26–45. https://doi.org/10.1016/j.sleep.2016.03.019
Rolland, Y., Lauwers-Cances, V., Cristini, C., Abellan van Kan, G., Janssen, I., Morley, J.E., & Vellas, B. (2009). Difficulties with physical function associated with obesity, sarcopenia, and sarcopenic-obesity in community-dwelling elderly women: The EPIDOS (EPIDemiologie de l’OSteoporose) study. American Journal of Clinical Nutrition, 89, 1895–1900. https://doi.org/10.3945/ajcn.2008.26950
Santos Iglesias, P., Sierra, J.C., Garcia, M., Martínez, A., Sánchez, A., & Tapia, I.M. (2009). Index of Sexual Satisfaction (ISS): A study on the reliability and validity. International Journal of Psychology and Psychological Therapy, 9, 259–273.
Schaap, L.A., Koster, A., & Visser, M. (2013). Adiposity, muscle mass, and muscle strength in relation to functional decline in older persons. Epidemiologic Reviews, 35, 51–65. https://doi.org/10.1093/epirev/mxs006
Seetharaman, S., Andel, R., McEvoy, C., Dahl Aslan, A.K., Finkel, D., & Pedersen, N.L. (2015). Blood glucose, diet-based glyce-mic load and cognitive aging among dementia-free older adults. Journals of Gerontology, 70, 471–479. https://doi.org/10.1093/gerona/glu135
Shao, S., Gill, A.A., Zahm, S.H., Jatoi, I., Shriver, C.D., McGlynn, K.A., & Zhu, K. (2018). Diabetes and overall survival among breast cancer patients in the U.S. military health system. Cancer Epidemiology, Biomarkers, and Prevention, 27, 50–57. https://doi.org/10.1158/1055-9965.EPI-17-0439
Sheng, J.Y., Sharma, D., Jerome, G., & Santa-Maria, C.A. (2019). Obese breast cancer patients and survivors: Management considerations. Oncology, 32(8), 410–417.
Singh, R., Teel, C., Sabus, C., McGinis, P., & Kluding, P. (2016). Fatigue in type 2 diabetes: Impact on quality of life and predictors. PLOS ONE, 11(11), e0165652. https://doi.org/10.1371/journal.pone.0165652
Smyth, C. (2012). The Pittsburgh Sleep Quality Index (PSQI). In M. Boltz (Ed.), Try this: Best practices in nursing care to older adults. New York, NY: The Hartford Institute for Geriatric Nursing, New York University, College of Nursing.
Spiegel, K., Tasali, E., Leproult, R., & Van Cauter, E. (2009). Effects of poor and short sleep on glucose metabolism and obesity risk. Nature Reviews. Endocrinology, 5(5), 253–261. https://doi.org/10.1038/nrendo.2009.23
Spielberger, C.D., Gorsuch, R.L., & Lushene, R.E. (1970). Manual for the state-trait anxiety inventory (self-evaluation questionnaire). Palo Alto, CA: Consulting Psychologists Press.
Spielberger, C.D., Gorsuch, R.L., Lushene, R., Vagg, P.R., & Jacobs, G.A. (1983). Manual for the State-Trait Anxiety Inventory. Palo Alto, CA: Consulting Psychologists Press.
Srokowski, T.P., Fang, S., Hortobagyi, G.N., & Giordano, S.H. (2009). Impact of diabetes mellitus on complications and out-comes of adjuvant chemotherapy in older patients with breast cancer. Journal of Clinical Oncology, 27, 2170–2176. https://doi.org/10.1200/JCO.2008.17.5935
Tang, Z., Wang, J., Shang, H., Sun, L., Tang, F., Deng, Q., & Yu J. (2016). Associations between diabetes and quality of life among breast cancer survivors. PLOS ONE, 11(6), e0157791. https://doi.org/10.1371/journal.pone.0157791
Vissers, P.A., Falzon, L., va de Poll-Franse, L.V., Pouwer, F., & Thong, M.S. (2016). The impact of having both cancer and diabetes on patient-reported outcomes: A systematic review and directions for future research. Journal of Cancer Survivorship, 10, 406–415. https://doi.org/10.1007/s11764-015-0486-3
Vissers, P.A., Thong, M.S., Pouwer, F., den Oudsten, B.L., Nieuwenhuijzen, G.A., & von de Poll-Franse, L.V. (2014). The individual and combined effect of colorectal cancer and diabetes on health-related quality of life and sexual functioning: Results from the PROFILES registry. Supportive Care in Can-cer, 22, 3071–3079. https://doi.org/10.1007/s00520-014-2292-2
Von Ah, D., Habermann, B., Carpenter, J.S., & Schneider, B.L. (2013). Impact of perceived cognitive impairment in breast cancer survivors. European Journal of Oncology Nursing, 17, 236–241. https://doi.org/10.1016/j.ejon.2012.06.002
Von Ah, D., Harvison, K.W., Monahan, P.O., Moser, L.R., Zhao, Q., Carpenter, J.S., . . . Unverzagt, F.W. (2009). Cognitive function in breast cancer survivors compared to healthy age- and education-matched women. Clinical Neuropsychologist, 23, 661–674. https://doi.org/10.1080/13854040802541439
Von Ah, D., Storey, S., Crouch, A., Johns, S.A., Dodson, J., & Dutkevitch, S. (2016). Relationship of self-reported attentional fatigue to perceived work ability in breast cancer survivors. Cancer Nursing, 40, 464–470. https://doi.org/10.1097/NCC.0000000000000444
Von Ah, D., Storey, S., Tallman, E., Nielsen, A., Johns, S.A., & Pressler, S. (2016). Cancer, cognitive impairment, and work-related outcomes: An integrative review. Oncology Nursing Forum, 43, 602–616. https://doi.org/10.1188/16.ONF.602-616
World Health Organization. (2017). Body mass index—BMI. Retrieved from http://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a…
Yellen, S.B., Cella, D.F., Webster, K., Blendowski, C., & Kaplan, E. (1997). Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. Journal of Pain and Symptom Management, 13, 63–74.
Young-Hyman, D., de Groot, M., Hill-Briggs, F., Gonzalez, J.S., Hood, K., & Peyrot, M. (2016). Psychosocial care for people with diabetes: A position statement of the American Diabetes Association. Diabetes Care, 39, 2126–2140. https://doi.org/10.2337/dc16-2053
Zhou, Y., Zhang, X., Gu, C., & Xia, J. (2015). Influence of diabetes mellitus on mortality in breast cancer patients. ANZ Journal of Surgery, 85, 972–978. https://doi.org/10.1111/ans.12877
Zhu, B., Quinn, L., Kapella, M.C., Bronas, U.G., Collins, E.G., Ruggiero, L., . . . Fritschi, C. (2018). Relationship between sleep disturbance and self-care in adults with type 2 diabetes. Acta Diabetologica, 55, 963–970. https://doi.org/10.1007/s00592-018-1181-4
Zilliox, L.A., Chadreasekaran, K., Kwan, J.Y., & Russell, J.W. (2016). Diabetes and cognitive impairment. Current Diabetes Reports, 16(9), 87. https://doi.org/10.1007/s11892-016-0775-x




