Exercise Interventions to Manage Fatigue in Women With Gynecologic Cancer: A Systematic Review
Problem Identification: Fatigue has a negative impact on the quality of life of patients with cancer. The aim of this review is to evaluate studies on the effectiveness of exercise interventions in reducing fatigue in women with gynecologic cancer.
Literature Search: The review was conducted according to the PRISMA guidelines using the CINAHL®, MEDLINE®, EMBASE, PsycINFO®, and Cochrane Library databases. The Critical Appraisal Skills Programme was used for quality assessment.
Data Evaluation: Five studies met the inclusion criteria: three randomized, controlled trials and two single-arm trials.
Synthesis: Evidence suggests that exercise interventions result in significant reductions in fatigue in women with gynecologic cancer. However, the current evidence is limited. Additional studies are required to address the dose-dependent outcomes of exercise interventions on fatigue in women with gynecologic cancer.
Implications for Nursing: Findings support the positive effects of exercise interventions in reducing fatigue in women with gynecologic cancer, suggesting that healthcare professionals may consider including exercise programs into management plans for this population.
Jump to a section
A gynecologic cancer diagnosis affects multiple aspects of a woman’s life, including physical and psychological health status. Gynecologic cancer refers to a malignant condition of the female reproductive system which may include ovary, vulva, vagina, cervix, uterus (womb/endometrial), uterine tube(s), and placenta (Gupta & Rajwanshi, 2013). Cervical cancer is the fourth most common cancer in women, and the seventh most common overall cancer (Ferlay et al., 2015). Fitzmaurice et al. (2015) suggested that 1 in 70 women are expected to develop cervical cancer from the time of their birth to age 79 years.
The stage of the cancer and the management options available have significant affects on survival rates. For example, the survival rate of ovarian cancer during a 10-year period is 35%, and more than 55% of the cases are diagnosed at stage III or IV (Macdonald & Palmer, 2014). In the case of endometrial cancer, however, the survival rate during a 10-year period is 78% and the majority of patients are diagnosed at an early stage of the disease (Cancer Research UK, 2015). In the United States, the five-year survival rate is 84% (American Cancer Society, 2018).
Fatigue is a particularly common and troublesome symptom that may affect daily activity levels and negatively alter the quality of life of people diagnosed with cancer (Peters, Goedendorp, Verhagen, Bleijenberg, & van der Graaf, 2016). Cancer-related fatigue (CRF) is defined as “a disturbing, persistent, subjective sense of physical, emotional, and/or cognitive fatigue or exhaustion related to cancer or cancer treatment that is not proportional to recent activity and interferes with usual functioning” (Berger et al., 2015, p. 1,012). De Raaf, de Klerk, Timman, Hinz, and van der Rijt (2012) reported that most patients with cancer describe fatigue as being the most distressing symptom affecting their quality of life, including fatigue occurring during the treatment of women with gynecologic cancer. The identification and management of CRF is a key objective of the holistic assessment and management of the individual diagnosed with cancer. Identification of CRF and the development of interventions to ameliorate it may help patients to improve their overall quality of life.
The prevalence of fatigue among patients with cancer is estimated to be 50%–90% (Campos, Hassan, Riechelmann, & Del Giglio, 2011), depending on the sample size and recorded variables. A systematic review completed by Harrington, Hansen, Moskowitz, Todd, and Feuerstein (2010) indicated that fatigue in gynecologic cancer survivors ranked as the fourth most common symptom, affecting 17%–33% of women an average of three to eight years after diagnosis. Similar results have been observed in other studies (Beesley et al., 2008; Fitch & Steele, 2010).
CRF can be complex and can interact with a number of physiologic and psychological mechanisms. Two key theories exist regarding the underlying mechanisms of CRF: peripheral (muscular) and central (neural). Peripheral fatigue causes muscular decline and reduction in physical performance through effects on muscle excitation/contraction mechanisms caused by metabolic changes within the muscle (Ryan et al., 2007). On the other hand, central fatigue is linked to progressive failure to transmit motor neuron impulses (Meeusen, Watson, Hasegawa, Roelands, & Piacentini, 2006). Therefore, to reduce fatigue, individuals with cancer are advised to increase physical activity levels. Several organizations have published guidelines that recommend exercise programs to improve functional capacity and muscular strength and to reduce CRF, including the American College of Sports Medicine (Schmitz et al., 2010), the American Cancer Society (Rock et al., 2012), and the National Institute for Health and Care Excellence ([NICE], 2017). Despite the existence of guidelines to manage fatigue for all patients with cancer, an evaluation of the evidence focused specifically on gynecologic cancer is needed. The aim of this review was to evaluate the effectiveness of exercise interventions in reducing fatigue in women with gynecologic cancer and to establish the parameters that have been investigated to date.
Methods
This systematic review was undertaken according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines (Moher, Liberati, Tetzlaff, & Altman, 2010), and the quality assessment was completed using the Critical Appraisal Skills Programme ([CASP], 2018). The review used two CASP checklists: randomized controlled trials (RCTs) (11 questions) and cohort studies (12 questions). In line with previous systematic reviews (Barnett, Barnett, Nathan, Van Cauwenberg, & Cerin, 2017; Kordzadeh, Liu, & Jayanthi, 2017; Pandey, Shetty, Hamilton, Bhattacharya, & Maheshwari, 2012), each study was assigned a score as an overall summary of quality. This incorporated three sections: validity of the results, reporting of the results, and results helping locally. Each question from the checklist was answered with “yes,” “no,” or “cannot tell.” A question answered with “yes” was allocated a score of 1, “cannot tell” a score of 0.5, and “no” a score of 0. The total points awarded indicated the overall quality of the study. When considering the RCTs (11 questions), a study was determined to be of low quality when the score was 0–4, of modest quality if the score was 5–7, and of high quality if the score was 8–11. When using the CASP (2018) checklist for cohort studies (12 questions), a study was considered to be of low quality when the score was 0–4, of modest quality if the score was 5–8, and of high-quality if the score was 8–12.
Search Strategy
Studies investigating exercise interventions in women with gynecologic cancer were obtained by searching the electronic databases CINAHL® (from 1937 to August 2017), MEDLINE® (from 1946 to August 2017), EMBASE (from 1980 to August 2017), PsycINFO® (from 1980 to August 2017), and the Cochrane Library (from 1980 to August 2017). Examples of search terms used were: activit*, cancer*, carcinoma*, endometr*, exercis*, fatigue, genital neoplasms, female, physical activit*. Footnote chasing and hand searching were used to identify additional studies. The footnote chasing strategy used other authors’ references cited in previous studies on the topic (Cooper, Hedges, & Valentine, 2009).
Selection Criteria
The review included the following study designs: pilot studies, feasibility studies, and RCTs. Protocol papers or conference abstracts were excluded. For a study to be included in the review, the following criteria had to be met: (a) patients aged 18 years or older, (b) diagnosed with any type of gynecologic cancer (uterus, cervix, vagina, ovary, vulva, fallopian tube, and placenta), (c) any stage of the disease, (d) published in English, (e) specifically tested a physical activity or exercise intervention, and (f) included at least one valid outcome in terms of a fatigue scale. Studies were excluded from the review if an exercise intervention was not prospectively tested or fatigue was not assessed using a standardized instrument. To determine eligibility for inclusion, two independent reviewers assessed the title and abstract of all papers that were identified from the search strategy, with any discrepancies being resolved by a third independent reviewer.
Results
Figure 1 displays the PRISMA diagram and the final selection of the studies, based on the inclusion criteria. Two hundred and forty-five papers were identified from the initial search. Of these, 231 studies were excluded during title and abstract screening because the sample did not include women with gynecologic cancer, 27 studies did not test an exercise intervention, and 62 did not assess fatigue using a standardized instrument. The full text of 14 articles was obtained, and another 9 articles were excluded. Two studies did not include gynecologic cancer, two studies did not include exercise in the intervention component, and five did not measure fatigue. Five studies were included in the review: two RCTs and three single-arm trials. 
Quality Assessment
The studies were assessed using two CASP checklist tools. The quality of the methodology of the two RCTs was assessed, and one trial was scored as being of high quality (Donnelly et al., 2011), whereas von Gruenigen et al. (2009) scored as being of only modest quality. The other three studies (Basen-Engquist et al., 2014; Mizrahi et al., 2015; Moonsammy et al., 2013) were assessed using the CASP checklist for cohort studies and scored as being of modest quality. Details of the quality assessment of the included studies are shown in Tables 1 and 2. 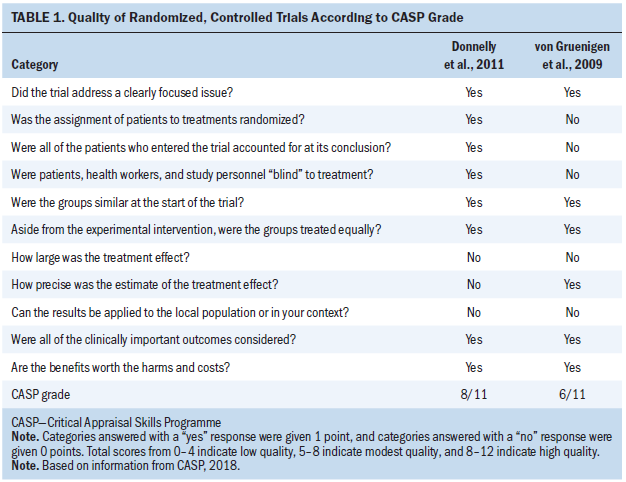
Study Design
The exercise studies included two RCTs (Donnelly et al., 2011; von Gruenigen et al., 2009) and three longitudinal, single-arm designs (Basen-Engquist et al., 2014; Mizrahi et al., 2015; Moonsammy et al., 2013). All participants, regardless of study design, were followed for at least six months. 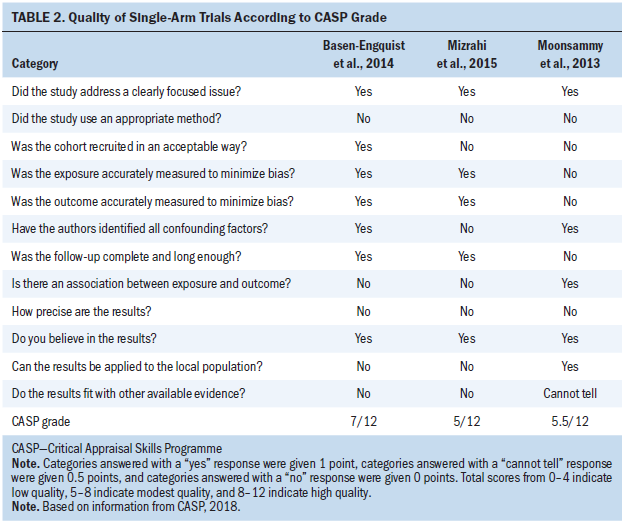
Of note, five of the studies reported “none” in the conflict of interest statement (Basen-Engquist et al., 2014; Donnelly et al., 2011; Mizrahi et al., 2015; Moonsammy et al., 2013; von Gruenigen et al., 2009). Failure to mention a conflict of interest could result in hidden study bias (Milton, 2012).
Participants’ Characteristics
The studies included women with ovarian, vulva, vaginal, cervix, uterus (womb/endometrial), uterine tube(s), or placental cancers at different stages of the disease and at different points of treatment. Table 3 summarizes the participants’ details. 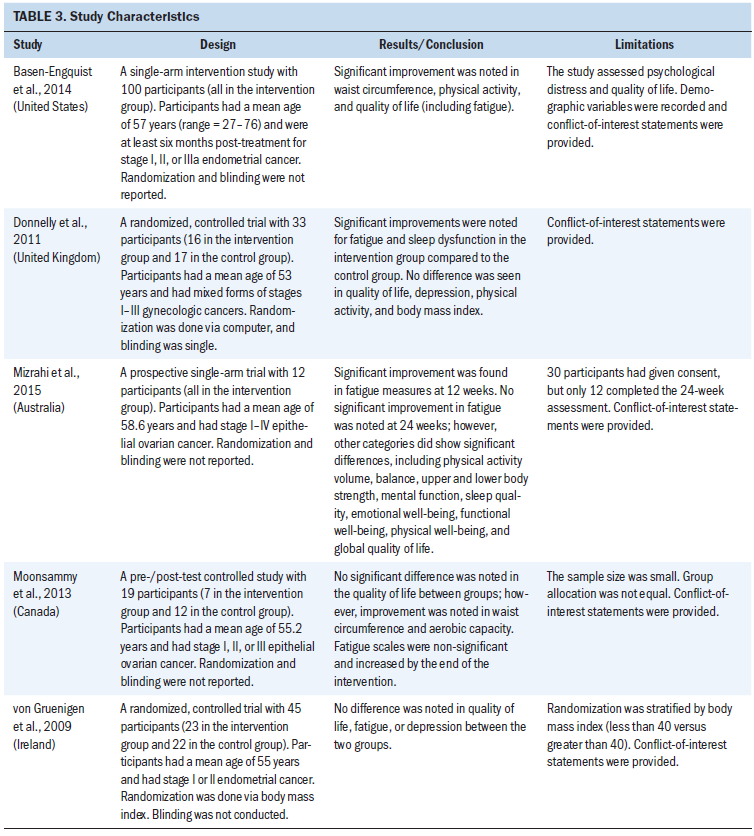
The five studies incorporated a total of 209 participants. Overall, the intervention groups included 163 patients, and the control groups included 46 patients. The differences between the numbers of women assigned to intervention versus control groups was primarily related to one of the single-arm studies enrolling 100 participants (Basen-Engquist et al., 2014).
Three studies involved participants diagnosed with cancer stages I–III (Basen-Engquist et al., 2014; Donnelly et al., 2011; Moonsammy et al., 2013); one study included stages I and II (von Gruenigen et al., 2009), while Mizrahi et al. (2015) included stages I–IV. The interventions in all studies were provided after surgery. Two studies involved participants during or after chemotherapy or radiation therapy (Basen-Engquist et al., 2014; Donnelly et al., 2011); two other studies (Mizrahi et al., 2015; Moonsammy et al., 2013) included participants after they had finished the second chemotherapy cycle. Only one study included participants who had completed treatment (von Gruenigen et al., 2009).
Instruments for Measuring Fatigue
A number of instruments were used to measure fatigue, including the Functional Assessment of Chronic Illness Therapy–Fatigue subscale (FACIT-F), the Multidimensional Fatigue Inventory, the vigor scale of the SF-36®, and fatigue subscales in the Quality of Life in Adult Cancer Survivors (QLACS) and the Somatic Psychological Health Report (SPHERE) scale. Three studies used the FACIT-F (Donnelly et al., 2011; Moonsammy et al., 2013; von Gruenigen et al., 2009). This unidimensional scale explores the experiences of fatigue on daily life. Donnelly et al. (2011) also used other scales, including the Multidimensional Fatigue Symptom Inventory–Short Form scale (MFSI-SF). Only one study measured fatigue by using the QLACS subscales (Basen-Engquist et al., 2014). Mizrahi et al. (2015) used the SPHERE scale to measure fatigue and the psychological dimensions of distress. In addition, three studies used the SF-36, which has four items in the form of energy/fatigue subscales (Basen-Engquist et al., 2014; Mizrahi et al., 2015; von Gruenigen et al., 2009).
Intervention Types
Most of the reviewed studies employed interval training and other forms of exercise. Despite the similarity in the exercise modality of the interventions, the intensity, frequency, duration, and the length of the exercises varied between studies. Table 4 provides a summary of the variations with regard to interventions in each study. The studies evaluated home-based exercise and behavior change interventions, or a combination of exercise and nutrition for six months (Basen-Engquist et al., 2014; Donnelly et al., 2011; Mizrahi et al., 2015; Moonsammy et al., 2013) and 12 months (von Gruenigen et al., 2009). The interventions aimed to meet particular requirements in terms of physical activity: 30 minutes for five days for a total of 150 minutes of physical activity per week. All studies commenced with an exercise counseling session. However, how contact was maintained with the participants varied throughout the studies. Three studies provided a booklet and a telephone follow-up every week for two months (Basen-Engquist et al., 2014; Donnelly et al., 2011; Mizrahi et al., 2015), whereas Moonsammy et al. (2013) conducted a follow-up call with participants every two weeks by telephone and von Gruenigen et al. (2009) conducted a follow-up discussion with participants only during the sessions. 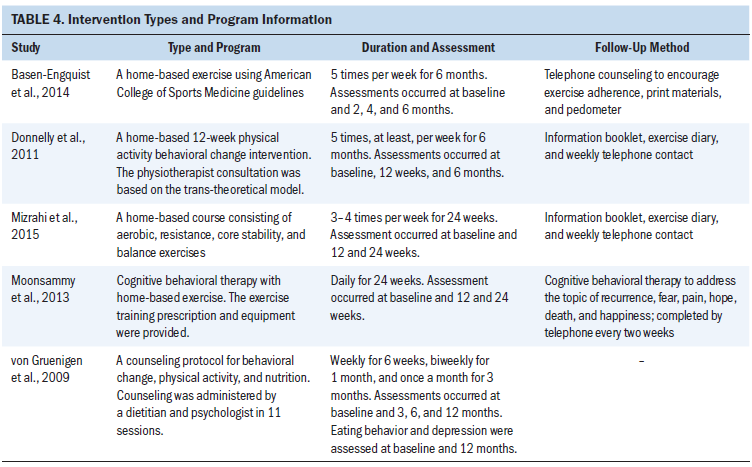
Effect of Exercise on Cancer-Related Fatigue
Three studies reported significant improvements in fatigue outcomes over time. The single-arm study conducted by Basen-Engquist et al. (2014) included endometrial cancer survivors (stage I, II, or IIIa; N = 100) and evaluated a six-month intervention consisting of home-based exercise. The study assessed psychological distress and quality of life at baseline and at six months. Weekly exercise, fitness, and anthropometrics were evaluated at baseline (n = 100), two months (n = 87), four months (n = 83), and six months (n = 79). The study used a computer to assess the home-based exercise. The participants wore medical-grade, activity-monitoring watches, which collected activity count data in 60-second intervals. Other recorded laboratory measures included waist circumference, cardiorespiratory fitness test, and breath-by-breath measurement of respiratory gas exchange to predict maximal oxygen uptake (VO2 max). The participants received printed materials on their physical activity goals, described as 30 minutes of exercise per day, five times per week. To encourage exercise adherence, telephone counseling was provided every week in the first two months, every two weeks in the third and fourth months of the study, and once a month during the fifth and sixth months. Fatigue was measured by the subscales of the QLACS, and significant improvement at the six-month assessment was found with p = 0.017. The study concluded that home-based exercise was beneficial for obese and non-obese endometrial cancer survivors. The study sample was large (N = 100), which increased its validity and reliability.
Mizrahi et al.’s (2015) single-arm study recruited 30 patients with ovarian cancer to evaluate the effectiveness of exercise interventions with supervision during the chemotherapy period. The 12-week intervention and assessment was completed by 21 participants (70% retention); only 12 participants completed the 24-week follow-up assessment (43% attrition rate).
The study intervention lasted 12 weeks, with 90 minutes of exercise per week. The intervention consisted of aerobic, core stability, resistance, and balance techniques. Aerobic exercises included swimming, walking, and cycling. Participants were given self-report physical activity diaries with the support of weekly telephone calls for monitoring adherence and progress. The study measured psychological and physiologic outcomes, with fatigue and psychological dimensions of distress assessed using SPHERE. The researchers found significant improvements at 12 weeks with regard to fatigue (p = 0.004), but this was not sustained at week 24. There were significant improvements in physical activity volume, upper and lower body strength, balance, lower body strength, sleep quality, mental function, functional well-being, emotional well-being, physical well-being, and global quality of life.
Donnelly et al. (2011) conducted an RCT with a total of 12 individuals allocated to an intervention group who received a home-based physical activity intervention. The control group consisted of 17 women who received standard care. This study assessed the feasibility and efficacy of a behavioral change physical activity intervention to manage CRF experienced by women with gynecologic cancer. The study included participants undergoing treatment and those who had completed treatment. The effects of the gynecologic cancer differed in terms of clinical features, risk factors, and the management and prognoses. The intervention program included walking and strengthening exercises to meet the requirement of 30 minutes of daily physical activity for five days per week. The participants received an initial consultation with a physiotherapist to explain the intervention, followed by a weekly telephone call for 10 weeks. The primary outcome with regard to fatigue was assessed using the MFSI-SF. Fatigue was assessed at three time points: baseline, 12 weeks, and 6 months. The study found statistically significant differences between the groups in terms of fatigue at 12 weeks (p = 0.046) and 6 months (p = 0.01). However, no significant improvements were noted in global quality of life or depression. Physical functioning activity showed significant increases in both groups (p < 0.001).
These three studies provide some evidence to suggest that home-based physical activity can result in improvements in fatigue for women with gynecologic cancer. However, in two of the studies, these effects were not maintained at follow-up.
Von Gruenigen et al.’s (2009) RCT examined the effect of a six-month lifestyle intervention that included physical activity among endometrial cancer (stages I–II) survivors. The participants were randomly assigned by their body mass index (BMI) to ensure equivalence between groups. Twenty-three patients (the intervention group) received a counseling protocol, which covered nutrition and exercise, and they met weekly for six weeks with a registered dietitian and psychologist. The control group (22 patients) received an information brochure explaining the benefits of proper nutrition and physical activity for patients with cancer. The study assessed the participants at baseline and at 3, 6, and 12 months. Fatigue was assessed using the FACIT-F scale. Quality of life, functional status, depression, eating behavior, self-efficacy, anthropometric data weight, waist circumference, and BMI were also assessed. There was no significant reduction in fatigue or global quality-of-life outcomes at 12 months. Participants in the exercise arm lost significantly more weight than those in the control arm (3.5 kg compared to 1.4 kg, p = 0.02). Interestingly, depression improved significantly in the control group compared to the intervention group (p = 0.02). The reason for this was unclear.
Using a pre/post-test controlled study format, Moonsammy et al. (2013) assessed the feasibility and benefits of a 24-week home-based exercise intervention involving 19 women with ovarian cancer. Seven people were assigned to the treatment group (patient on treatment) and 12 to the surveillance group (patient after treatment completion). The intervention comprised a training session in home-based exercise in both groups. This included aerobic exercise (brisk walking) and 10 resistance exercises; they were provided with individualized equipment consisting of a yoga mat, a stability ball, and resistance bands. Both groups received biweekly telephone-based cognitive behavioral therapy. Participants in both groups were encouraged to exercise for 30–60 minutes, three to five times per week. Descriptions of exercises and diaries to record daily activity were given to the participants. A cognitive behavioral therapy counseling session was delivered by phone every two weeks and addressed the topics of anxiety, pain, hope, happiness, cancer recurrence, and death in both groups.
Fatigue was measured using the FACIT-F and fitness by waist circumference, cardiorespiratory fitness test, and breath-by-breath measurement to predict maximal oxygen uptake (VO2 max). The assessments occurred at baseline, 12 weeks, and 24 weeks. Fatigue measures did not indicate any significant improvements at 24 weeks. However, aerobic fitness increased in both groups, and significant increases in body fat percentages and waist circumferences were seen in the intervention group. The authors postulated that such weight gain could have been a result of chemotherapy treatment-related factors. Only 13 participants completed the final assessment at 24 weeks (3 from the treatment group and 10 from the surveillance group).
The result of the two studies (Moonsammy et al., 2013; von Gruenigen et al., 2009) suggested that there was improvement in other measurements, but no improvement in fatigue. However, in both studies, the sample sizes were small. Both studies recommended that additional research should be conducted using larger sample sizes. The studies’ conclusions stated that there is a beneficial effect of exercise because it can increase the physical fitness of the participant.
Discussion
Fatigue is a prevalent and debilitating symptom among gynecologic cancer survivors; nevertheless, it can potentially be reversed by modifiable factors such as physical activity (Basen-Engquist et al., 2014; von Gruenigen et al., 2009). This systematic review aimed to identify the effectiveness of exercise interventions with regard to reducing fatigue in gynecologic cancer survivors. The findings suggest that exercise interventions result in significant reductions in fatigue. The challenges presented include the measurement of fatigue, variations in exercise intervention, and relatively high subject attrition rates.
This review found that, in three studies (Basen-Engquist et al., 2014; Donnelly et al., 2011; Mizrahi et al., 2015), exercise interventions significantly reduced fatigue and had a positive effect in terms of improving quality of life. In addition, exercise interventions can improve lifestyle and decrease BMI and waist circumference. However, those studies specifically included women with ovarian and endometrial cancer, whereas one study included all types of women with gynecologic cancer. Consequently, the findings cannot be generalized to all types of women with gynecologic cancer. The study with the largest sample size (Basen-Engquist et al., 2014), a single-arm intervention, showed significant improvement in fatigue and global quality of life. Mizrahi et al.’s (2015) study had a small sample size, and because 12 of 30 participants completed the 24-week assessment, the results from the study must be interpreted with caution. Another limitation of the studies was that the single-arm design could not rule out attrition bias. Despite this, two preliminary nonrandomized studies (Moonsammy et al., 2013; von Gruenigen et al., 2009) showed that weight loss can be achieved in such a way as to improve overall quality of life as well as to reduce fatigue. However, Moonsammy et al. (2013) noted attrition in the treatment group (four participants), which created an unequal group distribution and size. Transfer bias cannot be ruled out in the case of unequal group distribution. Nevertheless, this pilot study gives a platform for further direction with regard to studies intended to test this intervention in women with ovarian cancer.
Review of Studies
Exercise interventions did not result in significant reductions in fatigue in two studies (Moonsammy et al., 2013; von Gruenigen et al., 2009). These studies included only women with ovarian and endometrial cancer, and the type of participants may have an effect on the result. Sekse, Hufthammer, and Vika (2015) reported significant differences in fatigue between groups of women with gynecologic cancer according to their diagnoses. In addition, the sample sizes were small in both studies. In contrast, Smits et al. (2015) performed a review of eight studies to evaluate lifestyle interventions to improve the quality of life of endometrial and ovarian cancer survivors. The results suggested significant improvements in quality of life and fatigue reduction. However, Smits et al. (2015) did not include all types of gynecologic cancers. A meta-analysis assessing the effect of an exercise component for women with gynecologic cancer was conducted by Lin, Frawley, Denehy, Feil, and Granger (2016). The review included seven studies and concluded that no significant effects were found in terms of fatigue, depression, and global quality of life. Only the physical activity level and BMI appeared to improve among women with gynecologic cancer. However, the review did not assess fatigue as a primary outcome, and four of the studies in the review did not assess CRF.
Fatigue severity may vary according to underlying gynecologic cancer type diagnoses. Two studies involved women with endometrial cancer (Basen-Engquist et al., 2014; von Gruenigen et al., 2009) and two studies involved ovarian cancer survivors (Mizrahi et al., 2015; Moonsammy et al., 2013). Only one study had accepted a mixed gynecologic cancer survivors participant group (Donnelly et al., 2011). The heterogeneity of the participants within the included trials may influence fatigue outcomes. A cross-sectional study investigated the prevalence of fatigue on the part of 120 women with gynecologic cancer of various types (Sekse et al., 2015). Reports of fatigue were significantly different according to the diagnoses (p = 0.04), with the greatest degree of fatigue being reported in women with cervical cancer, followed by women with ovarian cancer. In addition, no differences were noted in reports of fatigue according to stages of cancer among women with gynecologic cancer. The effect of the gynecologic cancer differed in terms of clinical features, risk factors, management, and prognoses of the tumor. This highlights the challenges in working with this population and the need for large-scale international studies.
The timing of interventions relative to completion of cancer treatment (during or after cancer treatment) may affect fatigue intensity. Within the studies included in this review, the timing of the interventions was set to identify the difference between pre- and post-chemotherapy and/or radiation therapy. Chase et al. (2015), who evaluated toxicity before and after chemotherapy treatment, found an association between the treatment and fatigue. In addition, several researchers found the same result (Martin et al., 2014; Sun et al., 2013). A number of studies examined the impact of radiation therapy on fatigue and found an association with fatigue prior to starting radiation therapy (Nout et al., 2009), at the beginning of radiation therapy (Ahlberg, Ekman, & Gaston-Johansson, 2005), and during the first week of radiation therapy (Vistad, Fossa, Kristensen, & Dahl, 2007).
Evaluating the methodologic quality of the RCTs is important for interpreting results. Only two RCTs specified the method of randomization between the groups. Von Gruenigen et al. (2009) used randomized assigned stratification by BMI. Donnelly et al. (2011) used computer-generated random number tables to randomize between the groups. If the researcher allocates participants, or does not specify the method of randomization, it can increase the risk of bias and interfere with factors that affect the study outcomes (Kim & Shin, 2014). One of the RCTs (Donnelly et al., 2011) was single-blinded (the outcome assessor), whereas von Gruenigen et al. (2009) did not give any details about the blinding method used. Blinding is important because it avoids the potential of bias in terms of the expectancy effects of the researchers or the participants (Sedgwick, 2013). It is recommended that studies implement blinding (single or double) to limit such bias (Kahan, Doré, Murphy, & Jairath, 2016).
The incidence and the severity of CRF may be influenced by the age of the patients. The five studies included participants aged 53–58.6 years. Bifulco et al. (2012) found that women with gynecologic cancer younger than age 45 years were more affected by fatigue than older women. In addition, a longitudinal study included the follow-up of 144 women who had been diagnosed with gynecologic cancer two years previously (Chan et al., 2001) and revealed that younger patients’ quality-of-life scores were lower than those of older patients. However, several studies found that fatigue and quality of life worsens in older age, but this was in a patient population with mixed cancer types (Butt et al., 2010; Cella, Lai, Chang, Peterman, & Slavin, 2002). Therefore, an important direction for additional research would be to examine the relationship between exercise intervention and the reduction of fatigue among younger women with gynecologic cancer.
The study interventions in this review varied in terms of the intensity, frequency, duration, and the length of exercise regimen. A Cochrane review identified the techniques used to promote exercise interventions in cancer survivors (Bourke et al., 2013). The program should involve goal setting, prompting practice, self-monitoring, and participant engagement to attempt generalization of the behaviors learned. The exercise should also be supervised and be based on the individual’s capabilities (frequency, duration, repetitions, intensity, or set). Therefore, supervised individual or group sessions are needed, and there should be an exploration of home-based exercises to identify the best method of delivery for the intervention. Some of the studies in the review included variable components in terms of intervention, including exercise and nutrition, resulting in a significant change in fatigue and quality of life.
Identifying the host country of the exercise intervention studies in women with gynecologic cancer is important because cultural differences and the type of healthcare provision in each country may affect the results of the study. The exercise studies included in the review were conducted in different countries: the United States (Basen-Engquist et al., 2014), the United Kingdom (Donnelly et al., 2011), Australia (Mizrahi et al., 2015), Canada (Moonsammy et al., 2013), and Ireland (von Gruenigen et al., 2009). Several researchers suggested that personal and cultural values can affect the physical activity behaviors of women (Abbasi, 2014; Caperchione, Kolt, Tennent, & Mummery, 2011; Henderson & Ainsworth, 2003). For example, Spector, Battaglini, and Groff (2013) found that Hispanic/Latina women demonstrated greater perceived barriers to exercise compared to Caucasian and African American women, which may indicate sociocultural differences.
Implications for Practice and Research
Fatigue is a particularly common and troublesome symptom that may affect daily activity levels and negatively affect the quality of life among people diagnosed with cancer (Mehnert et al., 2018; Tang, Von Ah, & Fulton, 2018). This review was focused on exercise interventions and the effects on CRF. The findings of this review support a positive effect on fatigue levels following exercise interventions in women with gynecologic cancer.
Additional studies are needed to examine the effect of exercise interventions in women with gynecologic cancer. Ideally, studies should use a robust research design, with adequate sample size and appropriate control of all variables. In addition, researchers may consider exercise that is based on the individual’s capabilities (frequency, duration, repetitions, intensity). Furthermore, consideration should be given to the use of validated, psychometrically sound instruments to measure the multidimensional aspects of CRF.
The evidence in this article should help healthcare professionals to integrate exercise interventions into management plans for women with gynecologic cancer. However this needs careful grading and delivery by the healthcare professional. Fatigue is a complex symptom that commands additional research into an optimum management strategy for the individual person. 
Conclusion
Exercise intervention may significantly reduce fatigue and potentially improve the quality of life of gynecologic cancer survivors. However, current evidence is limited related to the available number of RCTs and cohort studies that have evaluated the effectiveness of exercise interventions on CRF. Consequently, further studies are required to address the dose-dependent response of exercise interventions on fatigue in populations with gynecologic cancer.
About the Author(s)
Mohammed Al Maqbali, RN, Dip. Admin., BSc(Hons), MSc, is a PhD candidate and Ciara Hughes, PhD, is a senior lecturer in the School of Health Sciences, both in the Institute of Nursing and Health Research at Ulster University in Newtownabbey, Belfast, United Kingdom; Lynn Dunwoody, PhD, AFBPsS, C.Psychol, FHEA, is a lecturer in health psychology in the Psychology Research Institute at Ulster University in Coleraine, Londonderry, United Kingdom; Jane P. Rankin, BSc(Hons), MSc, is a physiotherapy clinical lead in the Specialist Services Physiotherapy Department at the Belfast Health and Social Care Trust in the United Kingdom; Eileen D. Hacker, PhD, APN, AOCN®, FAAN, is a professor and department chair in the School of Nursing at Indiana University in Indianapolis; and Jackie Gracey, PhD, is a lecturer in the School of Health Sciences in the Institute of Nursing Research at Ulster University. This review was conducted as part of a PhD thesis, which was funded by the Ulster University Vice-Chancellor’s Research Scholarship. Al Maqbali completed the data collection, statistical support, and provided analysis. All authors contributed to the conceptualization and design and the manuscript preparation. Gracey can be reached at jh.gracey@ulster.ac.uk, with copy to ONFEditor@ons.org. (Submitted March 2018. Accepted July 26, 2018.)
References
Abbasi, I.N. (2014). Socio-cultural barriers to attaining recommended levels of physical activity among females: A review of literature. Quest, 66, 448–467.
Ahlberg, K., Ekman, T., & Gaston-Johansson, F. (2005). The experience of fatigue, other symptoms and global quality of life during radiotherapy for uterine cancer. International Journal of Nursing Studies, 42, 377–386. https://doi.org/10.1016/j.ijnurstu.2004.07.008
American Cancer Society. (2018). Cancer facts and figures, 2018. Atlanta, GA: Author.
Barnett, D.W., Barnett, A., Nathan, A., Van Cauwenberg, J., & Cerin, E. (2017). Built environmental correlates of older adults’ total physical activity and walking: A systematic review and meta-analysis. International Journal of Behavioral Nutrition and Physical Activity, 14, 103. https://doi.org/10.1186/s12966-017-0558-z
Basen-Engquist, K., Carmack, C., Brown, J., Jhingran, A., Baum, G., Song, J., . . . Lu, K.H. (2014). Response to an exercise intervention after endometrial cancer: Differences between obese and non-obese survivors. Gynecologic Oncology, 133, 48–55.
Beesley, V., Eakin, E., Steginga, S., Aitken, J., Dunn, J., & Battistutta, D. (2008). Unmet needs of gynaecological cancer survivors: Implications for developing community support services. Psycho-Oncology, 17, 392–400. https://doi.org/10.1002/pon.1249
Berger, A.M., Mooney, K., Alvarez-Perez, A., Breitbart, W.S., Carpenter, K.M., Cella, D., . . . Smith, C. (2015). Cancer-related fatigue, version 2.2015. Journal of the National Comprehensive Cancer Network, 13, 1012–1039. https://doi.org/10.6004/jnccn.2015.0116
Bifulco, G., De Rosa, N., Tornesello, M.L., Piccoli, R., Bertrando, A., Lavitola, G., . . . Nappi, C. (2012). Quality of life, lifestyle behavior and employment experience: A comparison between young and midlife survivors of gynecology early stage cancers. Gynecologic Oncology, 124, 444–451.
Bourke, L., Homer, K.E., Thaha, M.A., Steed, L., Rosario, D.J., Robb, K.A., . . . Taylor, S.J. (2013). Interventions for promoting habitual exercise in people living with and beyond cancer. Cochrane Database of Systematic Reviews, 9, CD010192. https://doi.org/10.1002/14651858.CD010192.pub2
Butt, Z., Rao, A.V., Lai, J.S., Abernethy, A.P., Rosenbloom, S.K., & Cella, D. (2010). Age-associated differences in fatigue among patients with cancer. Journal of Pain and Symptom Management, 40, 217–223. https://doi.org/10.1016/j.jpainsymman.2009.12.016
Campos, M.P., Hassan, B.J., Riechelmann, R., & Del Giglio, A. (2011). Cancer-related fatigue: A practical review. Annals of Oncology, 22, 1273–1279. https://doi.org/10.1093/annonc/mdq458
Cancer Research UK. (2015). Uterine cancer statistics. Retrieved from http://www.cancerresearchuk.org/health-professional/cancer-statistics/s…
Caperchione, C.M., Kolt, G.S., Tennent, R., & Mummery, W.K. (2011). Physical activity behaviours of culturally and linguistically diverse (CALD) women living in Australia: A qualitative study of socio-cultural influences. BMC Public Health, 11, 26. https://doi.org/10.1186/1471-2458-11-26
Cella, D., Lai, J.S., Chang, C.H., Peterman, A., & Slavin, M. (2002). Fatigue in cancer patients compared with fatigue in the general United States population. Cancer, 94, 528–538.
Chan, Y.M., Ngan, H.Y., Li, B.Y., Yip, A.M., Ng, T.Y., Lee, P.W., . . . Wong, L.C. (2001). A longitudinal study on quality of life after gynecologic cancer treatment. Gynecologic Oncology, 83, 10–19.
Chase, D.M., Kauderer, J., Wenzel, L., Ramondetta, L., Cella, D., Long, H.J., & Monk, B.J. (2015). Factors associated with grade 3-4 treatment-related toxicity in women with advanced or recurrent cervical cancer: An exploratory analysis of NRG Oncology/Gynecologic Oncology Group trials (COG) 179 and 204. International Journal of Gynecological Cancer, 25, 303–308.
Cooper, H., Hedges, L.V., & Valentine, J.C. (2009). The handbook of research synthesis and meta-analysis (2nd edition). New York, NY: Russell Sage Foundation.
Critical Appraisal Skills Programme. (2018). CASP appraisal checklists. Retrieved from https://casp-uk.net/casp-tools-checklists
de Raaf, P.J., de Klerk, C., Timman, R., Hinz, A., & van der Rijt, C.C. (2012). Differences in fatigue experiences among patients with advanced cancer, cancer survivors, and the general population. Journal of Pain and Symptom Management, 44, 823–830. https://doi.org/10.1016/j.jpainsymman.2011.12.279
Donnelly, C.M., Blaney, J.M., Lowe-Strong, A., Rankin, J.P., Campbell, A., McCrum-Gardner, E., & Gracey, J.H. (2011). A randomised controlled trial testing the feasibility and efficacy of a physical activity behavioural change intervention in managing fatigue with gynaecological cancer survivors. Gynecologic Oncology, 122, 618–624.
Ferlay, J., Soerjomataram, I., Dikshit, R., Eser, S., Mathers, C., Rebelo, M., . . . Bray, F. (2015). Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. International Journal of Cancer, 136, E359–E386.
Fitch, M.I., & Steele, R. (2010). Identifying supportive care needs of women with ovarian cancer. Canadian Oncology Nursing Journal, 20(2), 66–74.
Fitzmaurice, C., Dicker, D., Pain, A., Hamavid, H., Moradi-Lakeh, M., MacIntyre, M.F., . . . Naghavi, M. (2015). The global burden of cancer 2013. JAMA Oncology, 1, 505–527.
Gupta, N., & Rajwanshi, A. (2013). Gynecological cancers: Pathology and cytological methods for diagnosis of gynecological cancers. In M.K. Shetty, Breast and gynecological cancers (pp. 39–55). New York, NY: Springer.
Harrington, C.B., Hansen, J.A., Moskowitz, M., Todd, B.L., & Feuerstein, M. (2010). It’s not over when it’s over: Long-term symptoms in cancer survivors—A systematic review. International Journal of Psychiatry in Medicine, 40, 163–181.
Henderson, K.A., & Ainsworth, B.E. (2003). A synthesis of perceptions about physical activity among older African American and American Indian women. American Journal of Public Health, 93, 313–317.
Kahan, B.C., Doré, C.J., Murphy, M.F., & Jairath, V. (2016). Bias was reduced in an open-label trial through the removal of subjective elements from the outcome definition. Journal of Clinical Epidemiology, 77, 38–43.
Kim, J., & Shin, W. (2014). How to do random allocation (randomization). Clinics in Orthopedic Surgery, 6, 103–109.
Kordzadeh, A., Liu, M.O., & Jayanthi, N.V. (2017). Male infertility following inguinal hernia repair: A systematic review and pooled analysis. Hernia, 21, 1–7. https://doi.org/10.1007/s10029-016-1560-0
Lin, K.Y., Frawley, H.C., Denehy, L., Feil, D., & Granger, C.L. (2016). Exercise interventions for patients with gynaecological cancer: A systematic review and meta-analysis. Physiotherapy, 102, 309–319. https://doi.org/10.1016/j.physio.2016.02.006
Macdonald, M., & Palmer, J. (2014). Palliative care in gynaecological oncology. Obstetrics, Gynaecology and Reproductive Medicine, 24(12), 365–370. https://doi.org/10.1016/j.ogrm.2014.10.004
Martin, L.P., Sill, M., Shahin, M.S., Powell, M., DiSilvestro, P., Landrum, L.M., . . . Schilder, R.J. (2014). A phase II evaluation of AMG 102 (rilotumumab) in the treatment of persistent or recurrent epithelial ovarian, fallopian tube or primary peritoneal carcinoma: A Gynecologic Oncology Group study. Gynecologic Oncology, 132, 526–530. https://doi.org/10.1016/j.ygyno.2013.12.018
Meeusen, R., Watson, P., Hasegawa, H., Roelands, B., & Piacentini, M.F. (2006). Central fatigue: The serotonin hypothesis and beyond. Sports Medicine, 36, 881–909.
Mehnert, A., Hartung, T.J., Friedrich, M., Vehling, S., Brähler, E., Härter, M., . . . Faller, H. (2018). One in two cancer patients is significantly distressed: Prevalence and indicators of distress. Psycho-Oncology, 27, 75–82. https://doi.org/10.1002/pon.4464
Milton, C.L. (2012). Trust-mistrust: Conflicts of interest and nurse research. Nursing Science Quarterly, 25, 133–136. https://doi.org/10.1177/0894318412437961
Mizrahi, D., Broderick, C., Friedlander, M., Ryan, M., Harrison, M., Pumpa, K., & Naumann, F. (2015). An exercise intervention during chemotherapy for women with recurrent ovarian cancer: A feasibility study. International Journal of Gynecological Cancer, 25, 985–992. https://doi.org/10.1097/IGC.0000000000000460
Moher, D., Liberati, A., Tetzlaff, J., & Altman, D.G. (2010). Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. International Journal of Surgery, 8, 336–341. https://doi.org/10.1016/j.ijsu.2010.02.007
Moonsammy, S.H., Guglietti, C.L., Santa Mina, D., Ferguson, S., Kuk, J.L., Urowitz, S., . . . Ritvo, P. (2013). A pilot study of an exercise and cognitive behavioral therapy intervention for epithelial ovarian cancer patients. Journal of Ovarian Research, 6, 21. https://doi.org/10.1186/1757-2215-6-21
National Institute for Health and Care Excellence. (2017). Advanced breast cancer: Diagnosis and treatment. Retrieved from https://www.nice.org.uk/guidance/cg81
Nout, R.A., Putter, H., Jürgenliemk-Schulz, I.M., Jobsen, J.J., Lutgens, L.C., van der Steen-Banasik, E.M., . . . Creutzberg, C.L. (2009). Quality of life after pelvic radiotherapy or vaginal brachytherapy for endometrial cancer: First results of the randomized PORTEC-2 trial. Journal of Clinical Oncology, 27, 3547–3556. https://doi.org/10.1200/JCO.2008.20.2424
Pandey, S., Shetty, A., Hamilton, M., Bhattacharya, S., & Maheshwari, A. (2012). Obstetric and perinatal outcomes in singleton pregnancies resulting from IVF/ICSI: A systematic review and meta-analysis. Human Reproduction Update, 18, 485–503. https://doi.org/10.1093/humupd/dms018
Peters, M.E., Goedendorp, M.M., Verhagen, C.A., Bleijenberg, G., & van der Graaf, W.T. (2016). Fatigue and its associated psychosocial factors in cancer patients on active palliative treatment measured over time. Supportive Care in Cancer, 24, 1349–1355. https://doi.org/10.1007/s00520-015-2909-0
Rock, C.L., Doyle, C., Demark-Wahnefried, W., Meyerhardt, J., Courneya, K.S., Schwartz, A.L., . . . Gansler, T. (2012). Nutrition and physical activity guidelines for cancer survivors. CA: A Cancer Journal for Clinicians, 62, 242–274. https://doi.org/10.3322/caac.21142
Ryan, J.L., Carroll, J.K., Ryan, E.P., Mustian, K.M., Fiscella, K., & Morrow, G.R. (2007). Mechanisms of cancer-related fatigue. Oncologist, 12(Suppl. 1), 22–34.
Schmitz, K.H., Courneya, K.S., Matthews, C., Demark-Wahnefried, W., Galvão, D.A., Pinto, B.M., . . . Schwartz, A.L. (2010). American College of Sports Medicine roundtable on exercise guidelines for cancer survivors. Medicine and Science in Sports and Exercise, 42, 1409–1426.
Sedgwick, P. (2013). Allocation concealment versus blinding in randomised controlled trials. BMJ, 347, f5518. https://doi.org/10.1136/bmj.f5518
Sekse, R.J., Hufthammer, K.O., & Vika, M.E. (2015). Fatigue and quality of life in women treated for various types of gynaecological cancers: A cross-sectional study. Journal of Clinical Nursing, 24, 546–555. https://doi.org/10.1111/jocn.12647
Smits, A., Lopes, A., Das, N., Bekkers, R., Massuger, L., & Galaal, K. (2015). The effect of lifestyle interventions on the quality of life of gynaecological cancer survivors: A systematic review and meta-analysis. Gynecologic Oncology, 139, 546–552.
Spector, D., Battaglini, C., & Groff, D. (2013). Perceived exercise barriers and facilitators among ethnically diverse breast cancer survivors. Oncology Nursing Forum, 40, 472–480. https://doi.org/10.1188/13.ONF.472-480
Sun, V., Otis-Green, S., Morgan, R., Wakabayashi, M., Hakim, A., Callado, M.E., . . . Grant, M. (2013). Toxicities, complications, and clinical encounters during intraperitoneal chemotherapy in 17 women with ovarian cancer. European Journal of Oncology Nursing, 17, 375–380. https://doi.org/10.1016/j.ejon.2012.10.005
Tang, C.C., Von Ah, D., & Fulton, J.S. (2018). The symptom experience of patients with advanced pancreatic cancer: An integrative review. Cancer Nursing, 41, 33–44. https://doi.org/10.1097/NCC.0000000000000463
Vistad, I., Fossa, S.D., Kristensen, G.B., & Dahl, A.A. (2007). Chronic fatigue and its correlates in long-term survivors of cervical cancer treated with radiotherapy. BJOG, 114, 1150–1158. https://doi.org/10.1111/j.1471-0528.2007.01445.x
von Gruenigen, V.E., Gibbons, H.E., Kavanagh, M.B., Janata, J.W., Lerner, E., & Courneya, K.S. (2009). A randomized trial of a lifestyle intervention in obese endometrial cancer survivors: Quality of life outcomes and mediators of behavior change. Health and Quality of Life Outcomes, 7, 17. https://doi.org/10.1186/1477-7525-7-17




