“Just Living With Them”: Symptom Management Experiences of Rural Residents With Advanced Cancer
Purpose: To understand how rural residents with advanced cancer experience and manage their symptoms.
Participants & Setting: 16 adult patients with a diagnosis of advanced cancer, who were receiving antineoplastic treatment and living in rural areas of southeastern Iowa, participated in the study.
Methodologic Approach: Data were collected through semistructured, audio-recorded interviews using open-ended questions. Data were analyzed using content and dimensional analyses.
Findings: Four themes were developed from the completed interviews, including (a) barriers and challenges associated with rural cancer care, (b) physical symptoms experienced from the time of diagnosis through the cancer trajectory, (c) symptom management strategies used to control physical symptoms, and (d) perceptions of having cancer and the use of technology in managing symptoms.
Implications for Nursing: Rural residents with advanced cancer experience a wide range of physical symptoms that may affect their quality of life. Although residents may develop self-management strategies to cope with symptoms, additional guidance on and interventions for how best to manage physical symptoms are needed.
Jump to a section
Rural areas are experiencing a severe shortage of healthcare providers, with only one oncologist per 100,000 residents compared to five oncologists per 100,000 residents in urban and suburban areas (American Society of Clinical Oncology, 2017). Rural areas also often have reduced access to specialists, such as palliative care providers, psycho-oncologists, and oncology-certified staff (Hendren et al., 2011), which can lead to patients needing to travel long distances to see an oncologist (Baldwin et al., 2008). Patients with advanced cancer in rural areas are also more likely to receive highly aggressive treatment protocols (Moy et al., 2017; Singh, Williams, Siahpush, & Mulhollen, 2011). The combination of advanced disease and aggressive treatment can result in a high symptom burden among rural residents. According to previous studies, geographic rurality has contributed to a culture of stoicism and independence that can negatively influence the healthcare experiences of rural patients with cancer (Emery et al., 2013; Fischer et al., 2016).
Background
Patients with advanced cancer report an average of as many as 10 to 12 concurrent physical symptoms, which are associated with poorer functional status and quality of life and higher levels of distress (Eckerblad, Theander, Ekdahl, Jaarsma, & Hellstrom, 2015; Gilbertson-White et al., 2012). Symptom management interventions have largely been developed based on the experiences of people of all stages of disease (Berry et al., 2017; Cooley et al., 2017) or on cancer survivors who have completed treatment with curative intent (Freeman et al., 2015; Kapoor & Nambisan, 2018). In addition, the majority of published research on the symptom experience of patients with advanced cancer has primarily included urban and suburban residents who are receiving treatment at tertiary cancer care centers. Therefore, little is known about the symptom management experience of patients with advanced cancer living in rural areas or for those who are receiving care primarily at community or rural cancer centers. Although the existing literature describing rural–urban differences in the context of cancer care (e.g., use of breast-sparing surgeries, sentinel node utilization, use of definitive treatment for prostate cancer) is growing, studies on the management of physical symptoms are limited (Jacobs, Kelley, Rosson, Detrani, & Chang, 2008; Meden, St. John-Larkin, Hermes, & Sommerschield, 2002).
Significant improvements have been made in the management of physical cancer symptoms, resulting in a growing body of evidence-based interventions. Based on the existing literature, symptom scientists have begun to evaluate technology-enhanced symptom management interventions for patients with advanced cancer undergoing treatment (Donovan et al., 2014; Sherwood et al., 2005; Sikorskii et al., 2015; Yap et al., 2013). Sherwood et al. (2005) demonstrated that a telephone-based cognitive behavioral intervention could reduce symptom severity in people with advanced cancer. According to Sikorskii et al. (2015), delivering telephone-based interventions via nurses, health coaches, or automated call systems can achieve a significant reduction in symptom severity. Donovan et al. (2014) also tested a web-based symptom management intervention for women with ovarian cancer, which determined that participants in the intervention arm experienced lower symptom distress than those in the control arm. In addition, Yap et al. (2013) demonstrated that a pharmacist-delivered text-messaging intervention for the management of chemotherapy-induced nausea and vomiting is feasible and acceptable. However, no studies have been published on testing the efficacy of symptom management interventions in patients with advanced cancer living in rural areas (Gilbertson-White, Saeidzadeh, Yeung, Tykol, & Vikas, 2017). Researchers from the U.S. Department of Veterans Affairs have sought to use technology to improve symptom management outcomes among veterans with cancer. Silveira et al. (2011) found that a web-based, caregiver-focused symptom management intervention decreased veteran symptom severity and decreased caregiver burden. Researchers who used telehealth methods to deliver symptom management services to veterans with cancer found that these veterans had fewer chemotherapy-related hospitalizations compared to those who received normal controls (Chumbler et al., 2007). These web-based interventions were, however, not tailored for or evaluated in rural residents with advanced cancer.
Previous research on patients with cancer living in rural areas has focused on many issues, such as cognitive changes in post-treatment breast cancer survivors, spiritual beliefs of cancer survivors, and the support needs of African Americans with cancer (Hamilton, Powe, Pollard, Lee, & Felton, 2007; Meneses et al., 2018; Wenzel et al., 2012). Fewer studies have focused on patients with advanced cancer. Studies on palliative care for rural residents have demonstrated that a telephonic intervention (ENABLE) is feasible and efficacious in this population (Bakitas et al., 2015; Bakitas, Lyons, Hegel, Balan, Barnett, et al., 2009; Bakitas, Lyons, Hegel, Balan, Brokaw, et al., 2009); however, symptom management was the focus of only one session during a six-week intervention. The remaining sessions focused on other palliative care topics, such as problem solving, identifying resources, and communication. In addition, participants in the ENABLE studies were identified through tertiary care and academic medical centers and were not receiving their treatment from rural cancer clinics.
The U.S. Census Bureau defines urbanized areas as 50,000 people or more and urban clusters as 2,500 to 50,000 people (Ratcliffe, Burd, Holder, & Fields, 2016). The Federal Office of Rural Health Policy classifies all non-metropolitan counties as rural and uses Rural-Urban Commuting Area (RUCA) codes as an additional method of determining rurality (Larson & Skillman, 2005). Based on U.S. census data, a RUCA code is assigned to each census tract to account for distance to health services; tracts inside metropolitan counties with the codes 4 to 10 are considered rural (Larson & Skillman, 2005).
The U.S. health system is disjointed, particularly for individuals living in rural areas. If patients with cancer are not receiving coordinated care, it is unlikely that they are receiving comprehensive symptom management. Fragmented care and reduced access to specialists increase the burden on rural residents to understand their cancer treatments and symptoms that can occur as a result of treatment and to develop skills to self-manage symptoms. Therefore, the purpose of this study was to explore how patients with advanced cancer who live in rural areas experience and manage physical symptoms.
Methodologic Approach
This is a secondary analysis of a larger study that aimed to engage stakeholders for the development of a symptom management intervention for people with advanced cancer living in rural areas (Gilbertson-White et al., 2019). In the parent study, researchers used descriptions from participants with cancer about their diagnosis and symptoms they experienced, as well as how they managed their symptoms and used technology or the Internet, to better inform the development of symptom management interventions. In the current study, a qualitative descriptive approach was used to explore participants’ experiences with advanced cancer and cancer treatments (Lincoln & Guba, 1985; Willis, Sullivan-Bolyai, Knafl, & Cohen, 2016). The research team included two PhD-prepared nurse scientists, one PhD-prepared graduate assistant, and a medical oncologist.
Participants and Setting
This study was approved by the Human Subjects Committee of the University of Iowa’s institutional review board in Iowa City, Iowa. Purposive sampling was used to recruit men and women from three oncology centers in southeastern Iowa. The researchers aimed to interview men and women from diverse sociodemographic and clinical backgrounds to achieve a heterogeneous sample (Bachmann et al., 2009). The first recruitment site was a tertiary care hospital in a mid-sized city with a large rural population. The second and third recruitment sites were an outpatient medical oncology and a radiology outpatient clinic in two nonurban communities that serve large rural populations. All eligible participants had to have an advanced stage of cancer (stage III or IV) and live in a nonurban or rural area based on RUCA codes (Larson & Skillman, 2005).
Participants were screened for eligibility by oncology physicians and nurses working in the clinics and were introduced to the study by a clinical team member. Interested participants received additional information on the study from a member of the research team. All participants underwent an informed consent process and were reassured that their participation in the study would not affect their cancer care. One patient declined to participate after receiving more details from the research assistant, and another declined to participate between consent and data collection.
Data Collection
Consented participants were assigned a unique study identification number. Data were collected from September 2015 to May 2016. Trained research team members performed one-on-one interviews with participants (N = 16), averaging from 30 to 120 minutes, using a semistructured interview guide (see Figure 1). The interview guide consisted of open-ended questions with probes developed from the following predefined content areas: participant expectations following a cancer diagnosis, emotional responses related to a cancer diagnosis, physical problems participants experienced during their cancer experience, and use of technology and the Internet (general and specific to managing healthcare needs). Based on the participant’s preference, interviews were conducted at a convenient, private location within the clinic space or at the participant’s home and were audio recorded, with the interviewer taking observational notes to record the participant’s nonverbal cues (Carter, 1993). Participants received $25 upon completion of the interview. 
Demographic information, such as age, marital status, income level, highest level of education, and religious affiliation, was also collected. A demographic survey and a symptom burden measure were administered using an iPad following the interview process. The symptom burden measure was intentionally collected after the interview so that participants’ beliefs about symptoms were not influenced during the interview.
Data Analyses
Descriptive statistics, such as frequencies and means, were used to describe the demographic characteristics of the participant sample and to contextualize the qualitative analysis. All audio-recorded interview files were sent to a professional transcription company that transcribed each file verbatim into a Microsoft® Word document. Each transcript file was reviewed by a research team member and verified for accuracy with the audio recording. All interviews were uploaded into NVivo, version 10, to manage and organize the data.
Data analysis was conducted using a directed qualitative content approach (Hsieh & Shannon, 2005; Miles, Huberman, & Saldaña, 2014). The research team examined each interview transcript to label words, phrases, sentences, or paragraphs that participants shared that correlated with the predetermined content areas of interest (open coding). Information from family members or significant others who were present during the interviews, if requested by the participant, were recorded in the transcription, but the data were not included in the analysis. Open codes were compared and sorted into categories by the research team to discuss their impressions, systematically classify the codes, identify themes or patterns, and gain consensus on meaningful content. The analysis was conducted using a rigorous process that involved comparing the transcripts, codes, and categories to discern commonalities and differences.
To better understand certain concepts, such as characteristics of symptoms (Caron & Bowers, 2000), dimensional analysis was applied to themes to detect variations, specificity, and range (Schatzman, 1991). Matrices and hierarchical flowcharts were used to visualize relationships and patterns among themes and codes and to identify contrast across the dataset. Numerical counts were used to characterize the strength of the main themes and subthemes (Sandelowski, 2001). After each category was established, the research team reviewed the categories to summarize the key perspectives of the participants (Kwong et al., 2014). Interviews were conducted until no new themes or patterns were recognized. Participants from the third site were then recruited for additional sample diversity and to confirm findings.
The following steps were implemented to enhance the rigor of the research (Sandelowski, 1986, 1993): (a) the coinvestigator audited the data by performing dual coding and reviewing the data to ensure the credibility of the analysis, (b) three research team members discussed data exemplars, coding, and analytical decisions (Moser & Korstjens, 2018), and (c) all interview data and notes were documented using NVivo software to provide an audit trail of coding and analytical conditions.
Findings
Participants
Six men and 10 women completed single interviews. Participants were a mean age of 68.75 years, with a range of 40–81 years. The majority of participants self-identified as White (n = 15). Sample characteristics are summarized in Table 1. 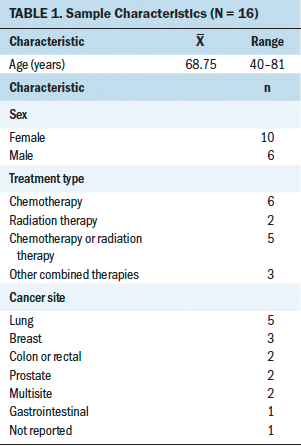
Qualitative Findings
The information provided by the participants explored additional topics outside of the four content areas that were established by the research team. To highlight how these findings relate to the symptoms experienced by rural residents who are surviving with advanced cancer, the following themes were developed:
• Barriers and challenges associated with living in a rural area and receiving care for cancer
• Physical symptoms that participants experienced from the time of diagnosis through the interview
• Management strategies that the participants used to control or mitigate physical symptoms
• Participant perceptions and use of technology in the context of having cancer and managing symptoms
These four themes represent the overarching experiences of the participants in this sample. Major themes, descriptions, and additional example quotes from participants are presented in Figure 2. Although emotional symptoms often occur at the same time as or are relative to physical symptoms, how participants described their emotional responses to and coping with a diagnosis of cancer did not fit within overall themes of symptom experience and management. 
Barriers and challenges: Participants described issues associated with living in a rural area that affected their receipt of oncology care. Most of the challenges were either financial concerns or logistical barriers. Financial concerns included treatment costs, as well as other financial issues, such as general financial stress and out-of-pocket expenses (e.g., transportation costs). Logistical barriers included driving long distances to see providers and the inconvenience of having their care spread out over a large area, particularly during inclement weather. One participant (male, aged 75 years) noted:
When them roads get icy and it’s dark in the morning, especially when you’re leaving . . . about 6 in the morning and all the incoming traffic’s there, it makes me nervous to drive in that stuff, but I can do it.
Another participant (male, aged 69 years) described the tiredness associated with long drives: “Right now, I don’t want to drive more than is necessary because it’s tiring. Not totally exhausting, but tiring.” However, they coped with it; the same participant stated, “Like I say, most of the stuff that I deal with, I’m able to deal with it.” Often, logistical barriers and financial issues were linked (e.g., hotel costs during overnight drives for next-day oncology visits).
Physical symptoms: Participants described the number of symptoms experienced, what they felt caused symptoms, and how possible drug–drug interactions of cancer treatments or medications to alleviate symptoms caused additional side effects. In addition, participants described their preexisting expectations of symptoms associated with cancer or cancer treatment, based on the experiences of their family members or friends. By far, reports of a wide range of physical symptoms dominated participant interviews. Participants described 53 total symptoms, which were categorized into 17 common symptom experiences. Gastrointestinal symptoms (n = 14), fatigue (n = 12), mucositis (n = 7), neurologic symptoms (n = 6), pain (n = 6), and sleep disturbances (n = 6) were the most frequently described physical symptoms. Participants reported the severity and impact of symptoms on activities of daily living or other daily activities.
When describing their symptoms, participants often included their perceptions of what caused the symptom (e.g., side effect of the treatment for another symptom), the patterns of symptom presentation, the frequency of symptoms, and their beliefs that symptoms could not be controlled. Participants often attributed the sources of their symptoms to various aspects of cancer (e.g., cancer treatments, medications, other symptoms). In this sample, treatments, such as chemotherapy and radiation therapy, were seen as the reasons participants experienced fatigue, sleep disturbances, and dizziness.
Treatments were believed to have caused symptoms because participants noticed altered patterns immediately following treatment. Noticing treatment patterns was an important aspect of participants’ developing an understanding of their symptoms and helping them to adjust to the symptom experience. Some participants were able to see interconnecting relationships between symptoms. One participant (male, aged 66 years) described the causes of different symptoms as, “Difficulty swallowing, all that kind of contributing overall to weight loss, and then I think that all contributing to low energy.” However, the interconnectedness of symptoms sometimes masked the source of the issue. One participant (male, aged 69 years) with swallowing difficulties said, “Well, most of it connected with the stomach and . . . I don’t know if it’s heartburn, acid reflux, indigestion. I’m not sure just how all those are tied together. I know they’re related.”
Based on the experiences of others, participants also had preexisting expectations about the symptoms that they would experience while receiving cancer treatment. One participant said, “[A friend] has an older sister who just got done with a bout of breast cancer. We compare notes.” Participants discussed the frequency with which they experienced highly distressing symptoms or, conversely, that they expected to experience symptoms but did not. One male participant stated, “I had some mild symptoms, but real mild, and I just didn’t really think a whole lot of it.” Some participants reported a complete lack of symptoms. One participant (male, aged 76 years) stated that, “I’ve had no side effects from this chemo whatsoever.” However, this same participant went on to share experiences that clinicians would consider to be side effects, although he did not discuss them routinely.
Management strategies: Participants described a wide range of techniques to help self-manage their symptoms. In addition, they shared their reflections on how well their symptom management strategies worked or did not work. One participant said, “I’m not sure that there is a strategy. More sleep?” Some participants articulated a trial and error process for determining the efficacy of their management strategies.
Behavioral interventions included asking for help, being proactive (e.g., being careful, standing after sitting for long periods of time), and advocating for oneself. One participant (male, aged 67 years) described a need for an individualized approach:
Just be proactive in relation to what side effects you do have because everybody has different side effects. You have to look at the one that you have and say, “OK, these are the ones that I’m experiencing. What can I do to lessen the effect of them?”
Pharmacologic interventions referred to the use of medications to manage cancer-related symptoms. Over-the-counter (e.g., diphenhydramine) and prescription medications (e.g., opioids) were described by participants as ways to manage anxiety, diarrhea, nausea, pain, esophageal reflux, skin concerns, and sleep disturbances. Medication was perceived as an easy and effective way to manage cancer-related symptoms; as one participant stated, “If I get diarrhea, take a pill. If I have a neck or backache, I’ll try and take some pain medicine for that or just try and treat whatever happens with whatever I have.” Another man said, “They told me if anything got too bad, I could call down here any time, and they’ll give me some medicine.” Other participants found that medications that were used to treat their symptoms created secondary symptoms, such as increased constipation from opioid medications for pain or feeling jittery because the dose of steroid premedication for chemotherapy was adjusted. Some participants described their process of taking medication to control physical symptoms. For example, one participant stressed the importance of developing a routine to ensure that he took his medicine at the same time every day and that doing so was in his best interest. Alternatively, one participant reported relying on memory only.
Participants also described many nonpharmacologic strategies for managing symptoms, such as physical activity. Participants reported using exercise, pacing activities, taking breaks, being careful not to overdo it, and recognizing the impact of inactivity on function. Other nonpharmacologic interventions included adjusting one’s diet, adjusting sleep habits, using heat or ice packs, and using reminders. One participant (female, aged 80 years) reported that she learned how to develop strategies to manage side effects by “just living with them.” Another participant (male, aged 67 years) described his experience with medication to manage sleeping difficulties:
It seems to be sort of like a learning experience, as you have more treatment and you see how you can manage what your body is doing and what things you can take that will help make it work better.
Others described practical strategies that they developed (e.g., carrying note cards to help with memory while experiencing cognitive impairments). In addition, several participants described monitoring activities that they used to help them manage their daily life with cancer. Monitoring activities included using a diary, writing things down, keeping records, and using a “hit or miss” approach to refining treatment dosages. One participant clearly described his understanding of how his treatment plan was based on his individual response to treatment, whereas another described understanding symptom presentation patterns based on his monitoring observations.
Perceptions and use of technology: When asked about the use of technology to manage their symptoms, participants reported using smartphones, tablets, and laptops or computers to access the Internet. Participants also described their experiences with technology (positive and negative), the barriers that they encountered, information that they were not willing to share over the Internet, and the use of their electronic health record portal for communicating with healthcare providers (i.e., MyChart). Several participants discussed browsing specific websites, such as WebMD or Google, for answers to their health-related questions. Discerning the validity of information on the Internet was an important aspect of seeking information. Participants often did not readily accept information and described the challenge of interpreting the validity of information found on the Internet.
Discussion
Rural residents living with advanced cancer diagnoses may experience challenges related to receiving cancer care, such as having a high physical symptom burden and needing to develop skills to self-manage their symptoms. The findings of this qualitative descriptive study can be used to develop symptom management interventions for rural residents with advanced cancer. In this study, rural residents with advanced cancer often reported self-care and continuous education throughout the cancer trajectory as strategies to manage symptoms without identifying or verbalizing symptoms or symptom management as such.
Because the focus of this study was to understand the symptom management experiences of people living in rural areas, the authors did not ask about access to providers or services. When participants reported challenges with access to care, it was not identified as an issue. Therefore, very little data about these barriers were collected from participants. Participants who received care at their local outpatient cancer center often traveled sizable distances from their homes for appointments with other providers on their healthcare team (e.g., surgical oncologists, radiation oncologists). Participants perceived significant financial and logistical barriers to receiving and managing cancer care as a result of their geographic location. This is consistent with previous research that indicates that rural residents experience significant barriers that affect the prevention, treatment, and surveillance of cancer (Jones et al., 2008; Rosenwasser et al., 2013; Scoggins et al., 2012). These barriers can lead to greater stress for patients in rural areas and can compound the development of distressing symptoms (Fitch et al., 2003). Participants in this study expressed feelings of anticipatory distress associated with having to travel to treatment centers and feelings of fatigue because of travel. Although logistical barriers did not prevent participants from seeking care, they increased the overall burden that rural residents faced while being treated for cancer.
In a study completed in rural Australia, patients with advanced cancer had moderate to high symptom management needs that went unaddressed (Rachakonda, George, Shafiei, & Oldmeadow, 2015). In addition, Gunn, Turnbull, McWha, Davies, and Olver (2013) found that even when rural residents seek psychosocial services from healthcare providers, the services are often not tailored to their needs. Although the health system in Australia differs from that of the United States, these findings were common to the participants in the current study’s sample as well.
Most participants denied experiencing symptoms when asked directly; however, they revealed their symptom experiences in subtle or indirect ways during the interviews. It is important to note the lack of identification or labeling of symptoms because patients with advanced cancer may not identify the physical changes or sensations they are experiencing as symptoms of their cancer, cancer treatments, or aging. Alternatively, patients may be aware of their symptoms but do not want to be perceived as a burden to healthcare providers, or they may be fearful of the consequences of revealing symptoms (Gunnarsdottir, Donovan, Serlin, Voge, & Ward, 2002; Heidrich et al., 2009; Ward et al., 1993). As first-line healthcare professionals, understanding the issues patients who are living with advanced cancer identify and how they take care of themselves is critical.
Based on the data collected, participants’ descriptions of symptoms and symptom management were not the same as the definition developed by the research team and clinicians. Frequently, participants indicated that they were “doing fine” and “not doing anything” to manage symptoms; however, further probing led to descriptions of significant symptoms experienced by the participants, often in the context of a story rather than in a response to a direct question. In addition, descriptions of symptoms and symptom management were discussed in response to other questions instead of to the questions intended to illicit these data. The researchers observed a process of participants denying symptoms when asked directly, but revealing symptom experiences when telling stories about or otherwise exploring their overall cancer experience. Midwestern stoicism is a strong cultural value that is often present in this demographic, which may lead to a lack of recognition of symptoms or an unwillingness to describe these experiences to researchers and clinicians for fear of being perceived as a complainer or bother (Moore, Grime, Campbell, & Richardson, 2013).
Participants reported that they managed symptoms using behavioral, pharmacologic, and nonpharmacologic strategies. During the interviews, the researchers purposefully did not use the phrase “symptom management” so as not to influence participants’ responses about their management strategies. Although the participants reported many strategies that they developed to cope with their symptoms, they did not identify these behaviors as managing symptoms. Overall, participants in this sample did not fully understand the concept of symptom management or have an understanding of the monitor and respond process for symptoms. Many participants did not use an intentional approach when managing their symptoms. Participants who were systematic in managing their symptoms stated that other aspects of their life (e.g., being an engineer) helped them to develop ways to analyze and respond to their cancer symptoms. However, this was often not the norm in this sample. Most participants used monitoring activities, not to necessarily manage their symptoms, but to manage daily life with cancer in general.
When participants were asked directly to describe their symptom management strategies, they overwhelmingly discussed the use of medication. This may be a result of information received from healthcare professionals. Participants indicated that conversations with medical professionals about symptoms often resulted in the receipt of a new medication; therefore, participants’ framing of symptom management may be limited to medication usage. Although participants engaged in problem solving and the use of nonmedication methods to manage symptoms, they did not view these behaviors as aspects of symptom management. Educating patients with advanced cancer on the definition of symptom management may be needed to ensure that patients develop an understanding of their role in mitigating symptoms. Previous research has indicated that symptom management interventions have resulted in an increase in health behaviors, improvement in health status, and increased self-efficacy (Lorig, Sobel, Ritter, Laurent, & Hobbs, 2001).
Almost universally, participants indicated that they or their family members used technology, such as the Internet, to learn about cancer or treatment side effects, which has been previously reported in the literature within the context of wanting to use reputable sources of knowledge to inform cancer care (Wickersham et al., 2014). Although experiences and comfort levels with technology and the Internet varied among participants, the majority noted that they or a family member would use technology for the purpose of managing their cancer and cancer symptoms.
Finally, this study addresses a gap in the literature on the experience of rural residents with cancer in general, as well as the symptom experience of patients with advanced cancer living in rural areas. The largest contributions of this research to the existing science are how the data relate to rural residents who are living with advanced cancer as opposed to survivors who were treated with curative intent and that rural residents with advanced cancer often educate themselves throughout the cancer trajectory to learn how to best self-manage their symptoms.
Limitations
This study only reflects the experience of patients with cancer in rural Iowa; therefore, the findings are not generalizable to other rural perspectives across the United States. Because members of the healthcare team first screened participants for eligibility before referring them to the research team, the potential for “gatekeeper bias,” in which having members outside of the research team assist with recruitment of qualitative samples can lead to bias, was present (Groger, Mayberry, & Straker, 1999). It is important to frame the findings of this study within the constraints of the sampling methods. In addition, data collection occurred primarily in a private area in the clinical setting, which may have inhibited the participants’ willingness to share certain experiences that they would not want their healthcare team to overhear. Although data collection was focused on understanding the symptom experience and participants’ ideas about symptom management, the word “symptom” was not used to limit bias from the participants. Asking the participants about symptom management directly may have influenced the findings.
Implications for Nursing
In this study, rural residents with advanced cancer experienced a range of physical symptoms that affected their daily lives. In addition to using standardized symptom assessment scales, oncology nurses can use open-ended questions to elicit more information on cancer symptoms from patients. When directly asked about symptoms, some patients may deny having any; however, asking overarching questions about what has been most troubling lately may encourage more detailed responses about the symptom management issues being experienced by patients who live in rural areas. Rural residents with advanced cancer may already be using highly effective symptom management strategies, but nurses can recommend resources for education or help patients to reconceptualize their behaviors so that they can recognize the symptom management strategies that work best for them. Nurses can also reinforce positive behaviors that patients are using to manage their symptoms. Helping patients to recognize what they are doing well in terms of symptom management can provide an enhanced sense of control and self-efficacy.
In addition to the symptoms that patients are actively experiencing, oncology nurses may assess patients’ individual approaches to symptom management, so that they can better recognize symptoms, the potential causes of those symptoms, and management strategies to mitigate symptoms. Systematic symptom management may develop naturally for some patients, but not for others. The evidence demonstrates that self-reporting of symptoms may improve median overall survival rates for patients with cancer receiving chemotherapy (Basch et al., 2017). Additional research is needed to evaluate how the Internet, mobile technology, and telemedicine can aid rural residents with cancer in reporting symptoms and developing symptom management skills for home care. Rural residents with cancer may be familiar with using technology and the Internet to find information on how to manage symptoms; however, they may need additional guidance on finding credible sites and using newer technologies. 
Conclusion
Rural residents with advanced cancer can have multiple barriers that may interfere with receiving information on symptom management and adhering to symptom management behaviors. Some individuals may not have a strong conceptualization of what symptom management is apart from taking medication. When people surviving with advanced cancer living in rural areas do engage in what healthcare professionals would consider to be symptom management, they often do not conceptualize it as such but rather as simply caring for themselves. Although there may be no difference between self-care activities and symptom management, it is important for healthcare professionals to be able to communicate well when developing, testing, delivering, and evaluating symptom management interventions for individuals with advanced cancer living in rural areas. Additional research is needed to better understand issues with symptom management delivery and care for rural residents with advanced cancer, as well as when to educate these individuals on how to best self-manage their symptoms.
About the Author(s)
Stephanie Gilbertson-White, PhD, APRN-BC, is an associate professor in the College of Nursing and Chi Yeung, MA, is a doctoral candidate in the College of Education, both at the University of Iowa in Iowa City; and Karen E. Wickersham, PhD, RN, is an assistant professor in the College of Nursing at the University of South Carolina in Columbia. This research was funded by grants from the Iowa Cancer Consortium (ICC 20142015), the College of Nursing at the University of Iowa (UIowaCON 2014–2016), and the National Palliative Care Research Center (NPCRC 2016–2018). Gilbertson-White and Wickersham contributed to the conceptualization and design. Gilbertson-White and Yeung completed the data collection. All authors provided the analysis and contributed to the manuscript preparation. Gilbertson-White can be reached at stephanie-gilbertson-white@uiowa.edu, with copy to ONFEditor@ons.org. (Submitted November 2018. Accepted December 21, 2018.)
References
American Society of Clinical Oncology. (2017). The state of cancer care in America, 2017: A report by the American Society of Clinical Oncology. Journal of Oncology Practice, 13, e353–e394. https://doi.org/10.1200/JOP.2016.020743
Bachmann, M.O., O’Brien, M., Husbands, C., Shreeve, A., Jones, N., Watson, J., . . . Mugford, M. (2009). Integrating children’s services in England: National evaluation of children’s trusts. Child, 35, 257–265. https://doi.org/10.1111/j.1365-2214.2008.00928.x
Bakitas, M., Lyons, K.D., Hegel, M.T., Balan, S., Barnett, K.N., Brokaw, F.C., . . . Ahles, T.A. (2009). The project ENABLE II randomized controlled trial to improve palliative care for rural patients with advanced cancer: Baseline findings, methodological challenges, and solutions. Palliatative and Supportive Care, 7, 75–86. https://doi.org/10.1017/s1478951509000108
Bakitas, M., Lyons, K.D., Hegel, M.T., Balan, S., Brokaw, F.C., Seville, J., . . . Ahles, T.A. (2009). Effects of a palliative care intervention on clinical outcomes in patients with advanced cancer: The Project ENABLE II randomized controlled trial. JAMA, 302, 741–749. https://doi.org/10.1001/jama.2009.1198
Bakitas, M.A., Tosteson, T.D., Li, Z., Lyons, K.D., Hull, J.G., Li, Z., . . . Ahles, T.A. (2015). Early versus delayed initiation of concurrent palliative oncology care: Patient outcomes in the ENABLE III randomized controlled trial. Journal of Clinical Oncology, 33, 1438–1445. https://doi.org/10.1200/JCO.2014.58.6362
Baldwin, L.M., Cai, Y., Larson, E.H., Dobie, S.A., Wright, G.E., Goodman, D.C., . . . Hart, L.G. (2008). Access to cancer services for rural colorectal cancer patients. Journal of Rural Health, 24, 390–399. https://doi.org/10.1111/j.1748-0361.2008.00186.x
Basch, E., Deal, A.M., Dueck, A.C., Scher, H.I., Kris, M.G., Hudis, C., & Schrag, D. (2017). Overall survival results of a trial assessing patient-reported outcomes for symptom monitoring during routine cancer treatment. JAMA, 318, 197–198. https://doi.org/10.1001/jama.2017.7156
Berry, D.L., Nayak, M.M., Abrahm, J.L., Braun, I., Rabin, M.S., & Cooley, M.E. (2017). Clinician perspectives on symptom and quality of life experiences of patients during cancer therapies: Implications for eHealth. Psycho-Oncology, 26, 1113–1119. https://doi.org/10.1002/pon.4455
Caron, C.D., & Bowers, B.J. (2000). Methods and application of dimensional analysis: A contribution to concept and knowledge development in nursing. In B.L. Rodgers, & K.A. Knafl (Eds.), Concept development in nursing: Foundations, techniques, and applications (2nd ed., pp. 285–319). Philadelphia, PA: W.B. Saunders.
Carter, B.J. (1993). Long-term survivors of breast cancer. A qualitative descriptive study. Cancer Nursing, 16, 354–361.
Chumbler, N.R., Kobb, R., Harris, L., Richardson, L.C., Darkins, A., Sberna, M., . . . Kreps, G.L. (2007). Healthcare utilization among veterans undergoing chemotherapy: The impact of a cancer care coordination/home-telehealth program. Journal of Ambulatory Care Management, 30, 308–317. https://doi.org/10.1097/01.JAC.0000290399.43543.2e
Cooley, M.E., Nayak, M.M., Abrahm, J.L., Braun, I.M., Rabin, M.S., Brzozowski, J., . . . Berry, D.L. (2017). Patient and caregiver perspectives on decision support for symptom and quality of life management during cancer treatment: Implications for eHealth. Psycho-Oncology, 26, 1105–1112. https://doi.org/10.1002/pon.4442
Donovan, H.S., Ward, S.E., Sereika, S.M., Knapp, J.E., Sherwood, P.R., Bender, C.M., . . . Ingel, R. (2014). Web-based symptom management for women with recurrent ovarian cancer: A pilot randomized controlled trial of the WRITE Symptoms intervention. Journal of Pain and Symptom Management, 47, 218–230. https://doi.org/10.1016/j.jpainsymman.2013.04.005
Eckerblad, J., Theander, K., Ekdahl, A., Jaarsma, T., & Hellstrom, I. (2015). To adjust and endure: A qualitative study of symptom burden in older people with multimorbidity. Applied Nursing Research, 28, 322–327. https://doi.org/10.1016/j.apnr.2015.03.008
Emery, J.D., Walter, F.M., Gray, V., Sinclair, C., Howting, D., Bulsara, M., . . . Holman, C.D. (2013). Diagnosing cancer in the bush: A mixed-methods study of symptom appraisal and help-seeking behaviour in people with cancer from rural Western Australia. Family Practice, 30, 294–301. https://doi.org/10.1093/fampra/cms087
Fischer, E.P., McSweeney, J.C., Wright, P., Cheney, A., Curran, G.M., Henderson, K., & Fortney, J.C. (2016). Overcoming barriers to sustained engagement in mental health care: Perspectives of rural veterans and providers. Journal of Rural Health, 32, 429–438. https://doi.org/10.1111/jrh.12203
Fitch, M.I., Gray, R.E., McGowan, T., Brunskill, I., Steggles, S., Sellick, S., . . . McLeese, D. (2003). Travelling for radiation cancer treatment: Patient perspectives. Psycho-Oncology, 12, 664–674. https://doi.org/10.1002/pon.682
Freeman, L.W., White, R., Ratcliff, C.G., Sutton, S., Stewart, M., Palmer, J.L., . . . Cohen, L. (2015). A randomized trial comparing live and telemedicine deliveries of an imagery-based behavioral intervention for breast cancer survivors: Reducing symptoms and barriers to care. Psycho-Oncology, 24, 910–918. https://doi.org/10.1002/pon.3656
Gilbertson-White, S., Aouizerat, B.E., Jahan, T., Paul, S.M., West, C., Schumacher, K., . . . Miaskowski, C. (2012). Determination of cutpoints for low and high number of symptoms in patients with advanced cancer. Journal of Palliative Medicine, 15, 1027–1036. https://doi.org/10.1089/jpm.2012.0045
Gilbertson-White, S., Saeidzadeh, S., Yeung, C.W., Tykol, H., & Vikas, P. (2017). Palliative and supportive interventions to improve patient-reported outcomes in rural residents with cancer. Journal of Community and Supportive Oncology, 15, e248–e255. https://doi.org/10.12788/jcso.0348
Gilbertson-White, S., Yeung, C.W., Saeidzadeh, S., Tykol, H., Vikas, P., & Cannon, A. (2019). Engaging stakeholders in the development of an eHealth intervention for cancer symptom management for rural residents. Journal of Rural Health, 35, 189–198. https://doi.org/10.1111/jrh.12297
Groger, L., Mayberry, P.S., & Straker, J.K. (1999). What we didn’t learn because of who would not talk to us. Qualitative Health Research, 9, 829–835. https://doi.org/10.1177/104973299129122180
Gunn, K., Turnbull, D., McWha, J.L., Davies, M., & Olver, I. (2013). Psychosocial service use: A qualitative exploration from the perspective of rural Australian cancer patients. Supportive Care in Cancer, 21, 2547–2555. https://doi.org/10.1007/s00520-013-1812-9
Gunnarsdottir, S., Donovan, H.S., Serlin, R.C., Voge, C., & Ward, S. (2002). Patient-related barriers to pain management: The Barriers Questionnaire II (BQ-II). Pain, 99, 385–396.
Hamilton, J.B., Powe, B.D., Pollard, A.B., 3rd, Lee, K.J., & Felton, A.M. (2007). Spirituality among African American cancer survivors: Having a personal relationship with God. Cancer Nursing, 30, 309–316. https://doi.org/10.1097/01.NCC.0000281730.17985.f5
Heidrich, S.M., Brown, R.L., Egan, J.J., Perez, O.A., Phelan, C.H., Yeom, H., & Ward, S.E. (2009). An individualized representational intervention to improve symptom management (IRIS) in older breast cancer survivors: Three pilot studies [Online exclusive]. Oncology Nursing Forum, 36, E133–E143. https://doi.org/10.1188/09.ONF.E133-E143
Hendren, S., Chin, N., Fisher, S., Winters, P., Griggs, J., Mohile, S., & Fiscella, K. (2011). Patients’ barriers to receipt of cancer care, and factors associated with needing more assistance from a patient navigator. Journal of the National Medical Association, 103, 701–710.
Hsieh, H.F., & Shannon, S.E. (2005). Three approaches to qualitative content analysis. Qualitative Health Research, 15, 1277–1288. https://doi.org/10.1177/1049732305276687
Jacobs, L.K., Kelley, K.A., Rosson, G.D., Detrani, M.E., & Chang, D.C. (2008). Disparities in urban and rural mastectomy populations: The effects of patient- and county-level factors on likelihood of receipt of mastectomy. Annals of Surgical Oncology, 15, 2644–2652. https://doi.org/10.1245/s10434-008-0053-5
Jones, A.P., Haynes, R., Sauerzapf, V., Crawford, S.M., Zhao, H., & Forman, D. (2008). Travel time to hospital and treatment for breast, colon, rectum, lung, ovary and prostate cancer. European Journal of Cancer, 44, 992–999. https://doi.org/10.1016/j.ejca.2008.02.001
Kapoor, A., & Nambisan, P. (2018). Usability and acceptance evaluation of ACESO: A Web-based breast cancer survivorship tool. Journal of Cancer Survivorship, 12, 316–325. https://doi.org/10.1007/s11764-017-0670-8
Kwong, J.P., Stokes, E.J., Posluns, E.C., Fitch, M.I., McAndrew, A., & Vandenbussche, K.A. (2014). The experiences of patients with advanced head and neck cancer with a percutaneous endoscopic gastrostomy tube: A qualitative descriptive study. Nutrition in Clinical Practice, 29, 526–533. https://doi.org/10.1177/0884533614532693
Larson, E., & Skillman, S. (2005). Rural urban commuting area code. Seattle, WA: WWAMI Rural Health Research Center, University of Washington.
Lincoln, Y.S., & Guba, E.G. (1985). Naturalistic inquiry. Newbury Park, CA: Sage.
Lorig, K.R., Sobel, D.S., Ritter, P.L., Laurent, D., & Hobbs, M. (2001). Effect of a self-management program on patients with chronic disease. Effective Clinical Practice, 4, 256–262.
Meden, T., St. John-Larkin, C., Hermes, D., & Sommerschield, S. (2002). Relationship between travel distance and utilization of breast cancer treatment in rural northern Michigan. JAMA, 287, 111. https://doi.org/10.1001/jama.287.1.111-JMS0102-5-1
Meneses, K., Benz, R., Bail, J.R., Vo, J.B., Triebel, K., Fazeli, P., . . . Vance, D.E. (2018). Speed of processing training in middle-aged and older breast cancer survivors (SOAR): Results of a randomized controlled pilot. Breast Cancer Research and Treatment, 168, 259–267. https://doi.org/10.1007/s10549-017-4564-2
Miles, M.B., Huberman, A.M., & Saldaña, J. (2014). Qualitative data analysis: A methods sourcebook (3rd ed.). Thousand Oaks, CA: Sage.
Moore, A., Grime, J., Campbell, P., & Richardson, J. (2013). Troubling stoicism: Sociocultural influences and applications to health and illness behaviour. Health, 17, 159–173. https://doi.org/10.1177/1363459312451179
Moser, A., & Korstjens, I. (2018). Series: Practical guidance to qualitative research. Part 3: Sampling, data collection and analysis. European Journal of General Practice, 24, 9–18. https://doi.org/10.1080/13814788.2017.1375091
Moy, E., Garcia, M.C., Bastian, B., Rossen, L.M., Ingram, D.D., Faul, M., . . . Iademarco, M.F. (2017). Leading causes of death in nonmetropolitan and metropolitan areas—United States, 1999–2014. Morbidity and Mortality Weekly Report, 66, 1–8. http://dx.doi.org/10.15585/mmwr.ss6601a1
Rachakonda, K., George, M., Shafiei, M., & Oldmeadow, C. (2015). Unmet supportive cancer care needs: An exploratory quantitative study in rural Australia. World Journal of Oncology, 6, 387–393. https://doi.org/10.14740/wjon928w
Ratcliffe, M., Burd, C., Holder, K., & Fields, A. (2016). Defining rural at the U.S. Census Bureau: American community survey and geography brief. Retrieved from https://www2.census.gov/geo/pdfs/reference/ua/Defining_Rural.pdf
Rosenwasser, L.A., McCall-Hosenfeld, J.S., Weisman, C.S., Hillemeier, M.M., Perry, A.N., & Chuang, C.H. (2013). Barriers to colorectal cancer screening among women in rural central Pennsylvania: Primary care physicians’ perspective. Rural and Remote Health, 13, 2504.
Sandelowski, M. (1986). The problem of rigor in qualitative research. Advances in Nursing Science, 8(3), 27–37.
Sandelowski, M. (1993). Rigor or rigor mortis: The problem of rigor in qualitative research revisited. Advances in Nursing Science, 16(2), 1–8.
Sandelowski, M. (2001). Real qualitative researchers do not count: The use of numbers in qualitative research. Research in Nursing and Health, 24, 230–240.
Schatzman, L. (1991). Dimensional analysis: Notes on an alternate approach to the grounding of theory in qualitative research. In A.L. Strauss, & D.R. Maines (Eds.), Social organization and social process: Essays in honor of Anselm Strauss (pp. 303–314). Hawthorne, NY: Aldine de Gruyter.
Scoggins, J.F., Fedorenko, C.R., Donahue, S.M., Buchwald, D., Blough, D.K., & Ramsey, S.D. (2012). Is distance to provider a barrier to care for Medicaid patients with breast, colorectal, or lung cancer? Journal of Rural Health, 28, 54–62. https://doi.org/10.1111/j.1748-0361.2011.00371.x
Sherwood, P., Given, B.A., Given, C.W., Champion, V.L., Doorenbos, A.Z., Azzouz, F., . . . Monahan, P.O. (2005). A cognitive behavioral intervention for symptom management in patients with advanced cancer. Oncology Nursing Forum, 32, 1190–1198. https://doi.org/10.1188/05.ONF.1190-1198
Sikorskii, A., Given, C.W., Siddiqi, A.E., Champion, V., McCorkle, R., Spoelstra, S.L., & Given, B.A. (2015). Testing the differential effects of symptom management interventions in cancer. Psycho-Oncology, 24, 25–32. https://doi.org/10.1002/pon.3555
Silveira, M.J., Given, C.W., Cease, K.B., Sikorskii, A., Given, B., Northouse, L.L., & Piette, J.D. (2011). Cancer carepartners: Improving patients’ symptom management by engaging informal caregivers. BMC Palliative Care, 10, 21. https://doi.org/10.1186/1472-684X-10-21
Singh, G.K., Williams, S.D., Siahpush, M., & Mulhollen, A. (2011). Socioeconomic, rural-urban, and racial inequalities in U.S. cancer mortality: Part I-all cancers and lung cancer and part II-colorectal, prostate, breast, and cervical cancers. Journal of Cancer Epidemiology, 2011, 107497.
Ward, S.E., Goldberg, N., Miller-McCauley, V., Mueller, C., Nolan, A., Pawlik-Plank, D., . . . Weissman, D.E. (1993). Patient-related barriers to management of cancer pain. Pain, 52, 319–324.
Wenzel, J., Jones, R.A., Klimmek, R., Krumm, S., Darrell, L.P., Song, D., . . . Ford, J.G. (2012). Cancer support and resource needs among African American older adults. Clinical Journal of Oncology Nursing, 16, 372–377. https://doi.org/10.1188/12.CJON.372-377
Wickersham, K.E., Happ, M.B., Bender, C.M., Engberg, S.J., Tarhini, A., & Erlen, J.A. (2014). Surviving with lung cancer: Medication-taking and oral targeted therapy. Geriatric Nursing, 35(2 Suppl.), S49–S56. https://doi.org/10.1016/j.gerinurse.2014.02.020
Willis, D.G., Sullivan-Bolyai, S., Knafl, K., & Cohen, M.Z. (2016). Distinguishing features and similarities between descriptive phenomenological and qualitative description research. Western Journal of Nursing Research, 38, 1185–1204. https://doi.org/10.1177/0193945916645499
Yap, K.Y., Low, H.X., Koh, K.S., Un, M., Shih, V., & Chan, A. (2013). Feasibility and acceptance of a pharmacist-run tele-oncology service for chemotherapy-induced nausea and vomiting in ambulatory cancer patients. Telemedicine Journal and e-Health, 19, 387–395. https://doi.org/10.1089/tmj.2012.0136

