Common and Co-Occurring Symptoms Experienced by Patients With Gastric Cancer
Problem Identification: Patients with gastric cancer experience multiple disease- and treatment-related symptoms. The purpose of this review was to describe the common and co-occurring symptoms experienced by patients with gastric cancer.
Literature Search: Search terms, such as gastric cancer, symptoms, and experience, were used to search PubMed®, CINAHL®, and PsycINFO® for empirical papers published from January 1990 to July 2019.
Data Evaluation: The search yielded 1,259 articles; 25 studies (21 observational and 4 interventional) were included in this review. Each study was systematically evaluated.
Synthesis: The most common symptoms were categorized into physical and affective/cognitive domains. Three to 17 (median = 7) symptoms occurred concurrently. The severity of most symptoms was reported as mild to moderate. However, patients experienced varying levels of symptom severity following treatment trajectories. Older age, female gender, advanced cancer stage, low socioeconomic status, and total gastrectomy were associated with a greater number and severity of symptoms.
Implications for Practice: Future research of symptom clusters may clarify the phenotypes and relationship between multiple co-occurring symptoms in patients with gastric cancer to develop targeted interventions that support symptom self-management for this population.
Jump to a section
Gastric (stomach) cancer was the fifth most frequently diagnosed cancer and the third leading cause of cancer deaths worldwide, with an estimated 1 million new cases and 783,000 deaths in 2018 (Bray et al., 2018). In the United States, an estimated 97,915 people were living with gastric cancer in 2015, with more than 26,240 estimated new cases and 10,800 estimated deaths in 2018 (Siegel et al., 2018). Patients diagnosed with gastric cancer have low five-year survival rates (10%−50%) because of advanced staging of disease at the time of diagnosis and the lack of effective treatments (Axon, 2006; Karimi et al., 2014; Venerito et al., 2018). People with gastric cancer experience multiple synergistic disease- and treatment-related symptoms. Investigators found that, on average, 10–15 of these symptoms occurred concurrently and included abdominal pain, weight loss, nausea, vomiting, dysphagia, dyspepsia, fatigue, and depression (Kim et al., 2016; Lee et al., 2016; Rausei et al., 2013). The undertreated symptoms can negatively influence patients’ health outcomes, such as functional performance, psychological status, quality of life, and survival rate (Kim et al., 2017; Konishi et al., 2016; Maconi et al., 2003; Rausei et al., 2013).
Symptom science is one of the four identified themes in the National Institute of Nursing Research (NINR, 2016) Strategic Plan, and it is an essential component of the research programs that are supported by NINR and the National Cancer Institute. Scientists and clinicians emphasized the importance of symptom management in cancer survivorship as a future cancer research priority in the United States (Jaffee et al., 2017). In addition, a new Symptom Science Center: A Resource for Precision Health was established by NINR (2019) in June; it emphasized the underlying mechanisms of multiple symptoms and developed personalized approaches for symptom management.
Understanding patients’ experience with co-occurring symptoms and their trajectories and predictors is critical to ensure appropriate assessment, education, and symptom management (Hockenberry et al., 2017; Miaskowski et al., 2004; National Institutes of Health State-of-the-Science Panel, 2004). For example, pain, fatigue, and sleep disturbance were experienced concurrently by breast cancer survivors and managed effectively through a mind–body intervention (Kwekkeboom et al., 2010). Multiple co-occurring symptoms have been identified in patients with breast (Bower, 2008; Tchen et al., 2003), lung (Cooley, 2000; Wong et al., 2017), prostate (Talcott et al., 2003), colorectal (Pettersson et al., 2014), and pancreatic (Burrell et al., 2018a, 2018b) cancers, and leukemia (Albrecht, 2014). However, there is limited evidence of the common symptoms (symptoms frequently occur) and co-occurring symptoms (symptoms occur at the same time) experienced by patients with gastric cancer, which is required to build symptom science in gastric cancer.
The purposes of this literature review were to describe the common and co-occurring symptoms experienced by patients with gastric cancer, and to synthesize symptom profiles (i.e., measurement, predictors, trajectories, and management) to create a comprehensive understanding of the state of the science on symptoms in patients with gastric cancer and inform future nursing research and practice for this population.
Methods
Literature Search
To understand the complexity of symptom science in patients with gastric cancer, the first author (Y.L.) conducted a comprehensive search of PubMed® (MEDLINE®), CINAHL®, and PsycINFO® for empirical papers published from January 1990 to July 2019. This date range was chosen because symptom-related studies in gastric cancer began to appear in 1990. Search terms stomach neoplasms, stomach, gastric, cancer, neoplasm, tumor, and tumour were combined with symptoms, symptoms and signs, symptom assessment, quality of life, and experience to elicit relevant literature. The article selection process is shown in Figure 1. 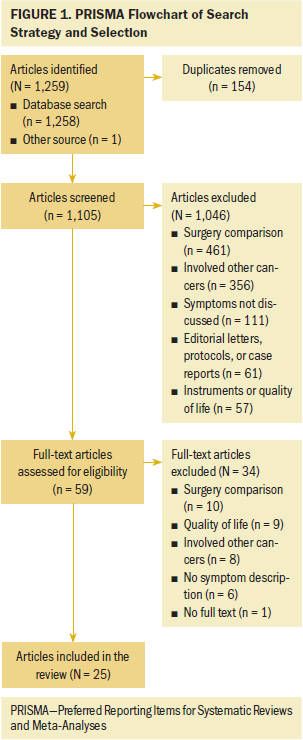
Inclusion and Exclusion Criteria
The inclusion criteria were as follows: studies involving symptoms experienced by people with gastric cancer; studies that considered the measurement, predictors, trajectories, and management of symptoms; reviews, systematic reviews, and meta-analyses; and published in English. The following were the exclusion criteria: editorial letters, comments, unpublished manuscripts, research protocols, and case reports; studies that did not report symptoms in their findings; studies that involved other cancer populations; studies that discussed only quality of life; and studies in which symptoms were a secondary outcome to a surgical procedure or medication trial (because these studies did not specifically describe patients’ experience with symptoms). The search yielded 1,259 primary research studies; 25 studies were included.
Data Evaluation
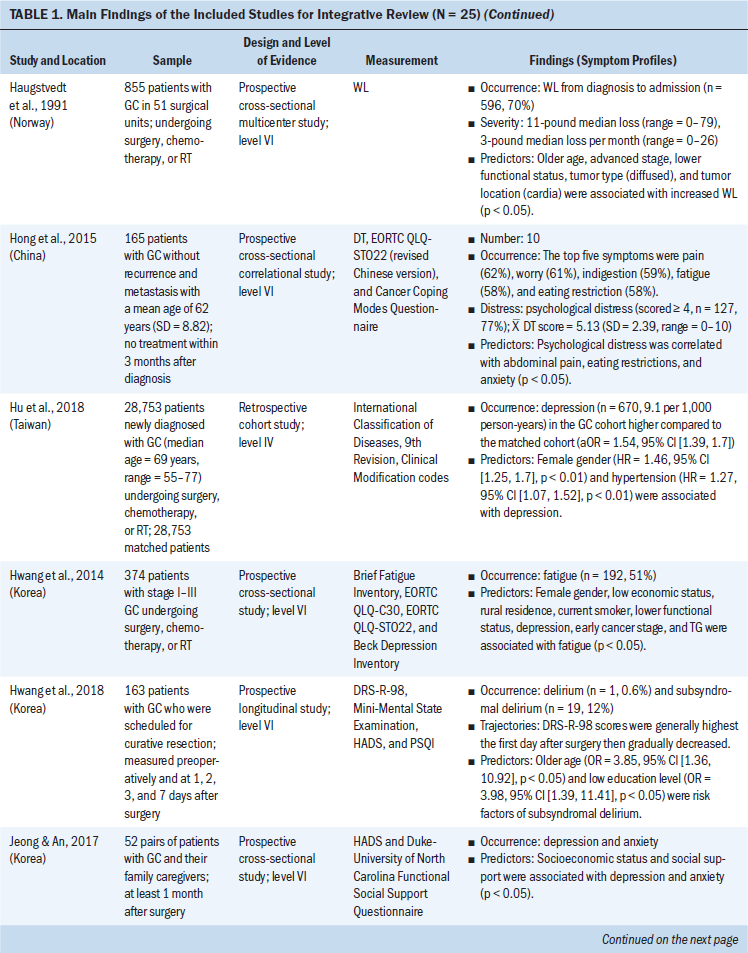
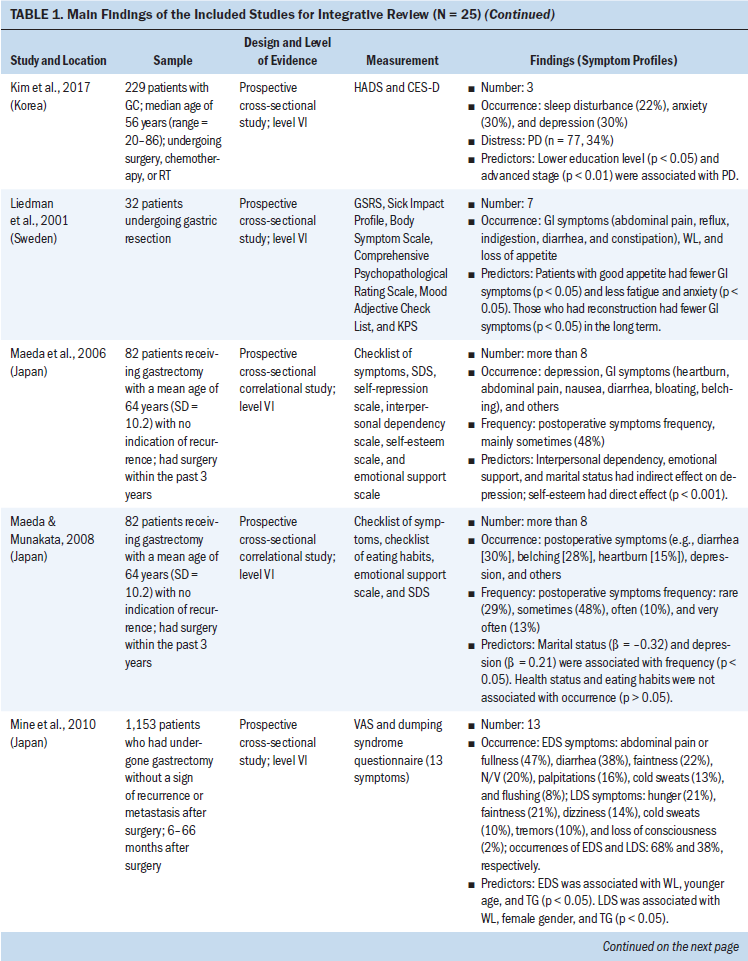
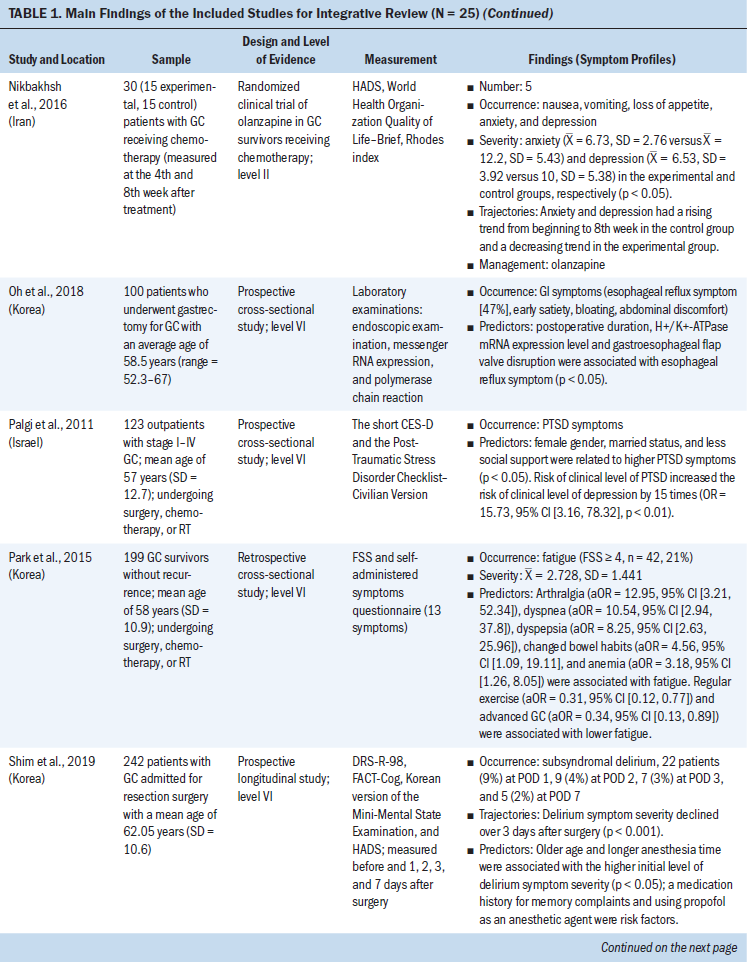
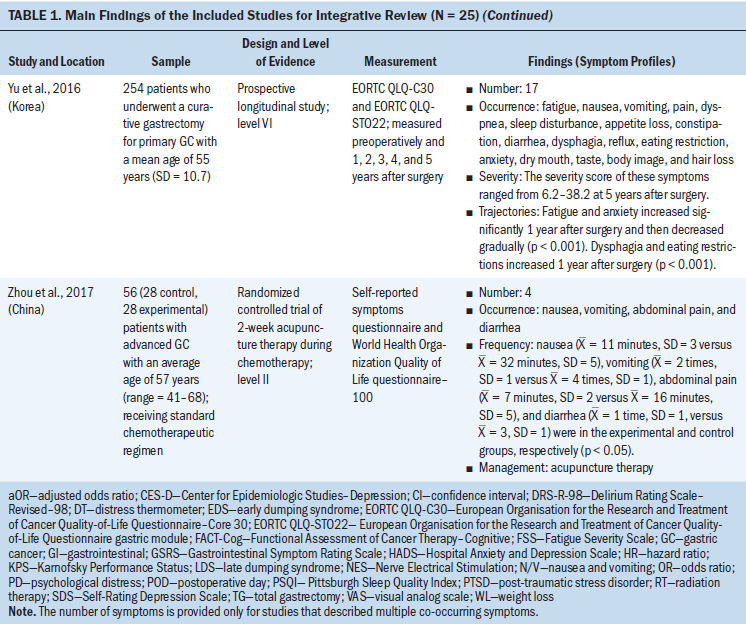
Data were evaluated using the hierarchy of evidence described by Melnyk and Fineout-Overholt (2011). The levels of evidence for this review consist of level II (high quality) to level VI (low quality); level II is evidence from well-designed randomized controlled trials, level III is evidence from well-designed controlled trials without randomization, level IV is evidence from well-designed case-control and cohort studies, and level VI is evidence from single descriptive or qualitative studies. For the included studies, the majority of studies (n = 20, 80%) were level VI, three were level II, one was level III, and one was level IV (see Table 1). The level of evidence for the body of literature was low quality. Data were extracted using the matrix method, which involves reading the documents, listing important issues, and selecting and adding column topics (Garrard, 2014). 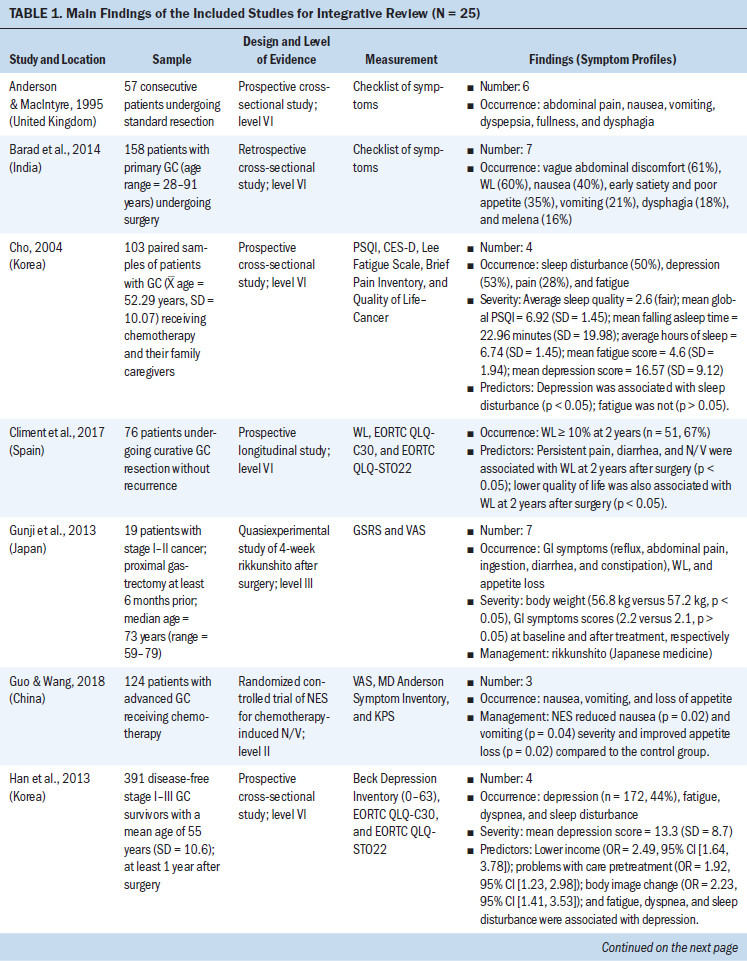
Results
Characteristics of the Studies
Twenty-five articles were included in this integrative review. All articles used quantitative methodologies; four articles were interventional studies, and the remainder (n = 21) were observational studies. Among the 20 quantitative descriptive studies, 16 reported cross-sectional data and 4 reported longitudinal data. One study used a cohort study design (Hu et al., 2018). The sample sizes ranged from 19 to 28,753 (median = 124). Studies originated from 11 countries or districts, including Korea (n = 10), Japan (n = 4), China (n = 3), Sweden (n = 1), Norway (n = 1), Spain (n = 1), United Kingdom (n = 1), Iran (n = 1), India (n = 1), Israel (n = 1), and Taiwan (n = 1). More than half (52%) of these studies were published in the past five years. Ten studies reported individual symptoms, and 15 reported multiple co-occurring symptoms.
Common and Co-Occurring Symptoms
The most frequent symptoms were categorized into physical symptoms (i.e., gastrointestinal [GI], fatigue, weight loss, and sleep disturbance), and affective/cognitive symptoms (i.e., anxiety, depression, post-traumatic stress disorder, and delirium). Patients with gastric cancer experienced multiple co-occurring symptoms at varying points before, during, and after treatment. Three to 17 symptoms (median = 7) were reported to occur concurrently in 15 studies.
Based on the Symptoms Experience Model (Armstrong, 2003), the studies were conceptually organized into four dimensions: occurrence, severity, frequency, and distress. All articles reported the occurrence of symptoms; seven studies reported symptom severity using different measures (Cho, 2004; Gunji et al., 2013; Han et al., 2013; Haugstvedt et al., 1991; Nikbakhsh et al., 2016; Park et al., 2015; Yu et al., 2016); three studies reported symptom frequency (Maeda et al., 2006; Maeda & Munakata, 2008; Zhou et al., 2017); and two articles assessed symptom distress (Hong et al., 2015; Kim et al., 2017). Not all study reports of common symptoms described all four dimensions of symptom experience; most reported one to two dimensions.
GI symptoms: GI symptoms were the most frequently occurring symptoms among gastric cancer survivors. Eight studies reported that patients experienced a wide range of GI symptoms. A retrospective study in a sample of 158 gastric cancer survivors showed that abdominal pain (61%) was the most common GI symptom reported by patients, followed by nausea (40%), early satiety and poor appetite (35%), vomiting (21%), dysphagia (18%), and melaena (16%) (Barad et al., 2014). Mine et al. (2010) conducted a cross-sectional study among 1,153 patients with gastric cancer and found that the occurrence of GI symptoms included abdominal pain or fullness (47%), diarrhea (38%), and nausea/vomiting (20%). Together, these results indicate that abdominal pain is the most prevalent of GI symptoms. Other common GI symptoms include nausea, vomiting, diarrhea, constipation, dysphagia, and reflux (Anderson & MacIntyre, 1995; Maeda et al., 2006; Maeda & Munakata, 2008; Oh et al., 2018; Zhou et al., 2017).
Fatigue: Three studies described fatigue among patients with gastric cancer. The prevalence ranged from 21% (Park et al., 2015) to 51% (Hwang et al., 2014). Park et al. (2015) reported that the mean fatigue severity score was 2.728 (SD = 1.441), as measured by the Fatigue Severity Scale (score range = 1–7), which indicated a moderate level of fatigue. In a study of 254 patients who underwent gastrectomy, Yu et al. (2016) reported mild levels of fatigue severity at baseline and five years after surgery. Therefore, the occurrence of fatigue was moderate and the severity was mild among patients with gastric cancer.
Weight loss: Weight loss was prevalent in patients with gastric cancer. The occurrence of weight loss was described in three studies as ranging from 60% (Barad et al., 2014; Climent et al., 2017) to 70% (Haugstvedt et al., 1991). In addition, Haugstvedt et al. (1991) reported a median weight loss of 11 pounds (range = 0–79) from the time of diagnosis through admission, and the median loss of weight was 3 pounds per month following treatment (range = 0–26). These studies indicate a high level of weight loss occurrence and severity in patients with gastric cancer.
Sleep disturbance: Disturbance in sleep was found to be problematic in two studies of patients with gastric cancer. The rate of sleep disturbance was reported as 22% in a sample of 229 patients with gastric cancer (Kim et al., 2017). In a study by Nikbakhsh et al. (2016), using the Pittsburgh Sleep Quality Index (PSQI), the average sleep quality was rated as 2.6 (fair sleep), the average hours slept were 6.74 hours (SD = 1.45), and the average time to fall asleep was 22.96 minutes (SD = 19.98). This suggests that patients with gastric cancer experienced sleep disturbance, with lower than the nationally recommended seven to nine hours of sleep and a longer time to fall asleep (Hirshkowitz et al., 2015).
Anxiety and depression: Anxiety and depression were the most prevalent affective symptoms experienced by patients with gastric cancer. Four studies described anxiety and depression. Hu et al. (2018) conducted a large-scale cohort study of 28,753 patients who were newly diagnosed with gastric cancer and found that depression among the gastric cancer cohort was 1.5 times higher than the matched control cohort (adjusted odds ratio [OR] = 1.54, 95% confidence interval [CI] [1.39, 1.7], p < 0.05). In a cross-sectional study of 229 gastric cancer survivors, Kim et al. (2017) reported anxiety (30%) and depression (30%) using a modified distress thermometer. The mean severity score of depression, assessed using the Beck Depression Inventory, was 13.3 (SD = 8.7), which indicated a mild level of severity (Han et al., 2013). Distress levels of anxiety and depression were reported to be as high as 77% in a study by Hong et al. (2015). In summary, these studies suggest that patients with gastric cancer experience anxiety and depression that is mild in severity but high in occurrence and distress.
PTSD: Cancer-related PTSD symptoms include feeling emotionally numb and feeling distant from other people (Palgi et al., 2011). In a cross-sectional study of 123 outpatients with stage I to III gastric cancer, investigators found an association between PTSD symptoms and depressive symptoms (r = 0.474, p = 0.001) (Palgi et al., 2011).
Delirium: Delirium was a neurocognitive symptom reported in two articles published by the same Korean research team. Hwang et al. (2018) reported that 19 patients (12%) with a mean age of 70.11 years (SD = 7.49) experienced postoperative subsyndromal delirium, measured by the Delirium Rating Scale-Revised-98 (DRS-R-98 = 8–14), and one patient (1%) experienced delirium (DRS-R-98 ≥ 15) in a sample of 163 participants; the severity of subsyndromal delirium was the highest on the first day after surgery and then gradually decreased. Shim et al. (2019) found that the occurrence of subsyndromal delirium was 9%, 4%, 3%, and 2% at 1, 2, 3, and 7 days after surgery, respectively, among 242 patients with gastric cancer (mean age = 62.05 years, SD = 10.6). The severity of subsyndromal delirium decreased at three days after surgery. Taken together, these results suggest that delirium is prevalent in those undergoing gastrectomy; its severity gradually decreased after surgery.
Synthesis of Symptom Profiles
The authors organized descriptions of the symptom profiles into four themes: symptom measurement, symptom predictors, symptom trajectories, and symptom management.
Symptom measurement: A range of instruments, with established validity and reliability, were used to measure symptoms in patients with gastric cancer. The most commonly used instruments for multiple co-occurring symptoms included the European Organisation for the Research and Treatment of Cancer Quality-of-Life Questionnaire–Core 30 (EORTC QLQ-C30) and its gastric module (EORTC QLQ-STO22), and the Gastrointestinal Symptom Rating Scale. The use of self-designed questionnaires, which lacked established validity and reliability, was reported in a small number of studies (Anderson & MacIntyre, 1995; Park et al., 2015; Zhou et al., 2017). The remaining studies used non–disease-specific symptom instruments (i.e., PSQI and DRS-R-98) with established validity and reliability to evaluate a single symptom experienced by patients with gastric cancer (Hwang et al., 2018; Kim et al., 2017).
Symptom predictors: Predictors of symptoms varied by studies and types of symptoms. Table 2 summarizes associations between different symptoms among patients with gastric cancer. Older age, female gender, low socioeconomic status (SES), low social support, advanced cancer stage, and total gastrectomy were associated with a higher number and severity of symptoms. Older age was associated with weight loss (Haugstvedt et al., 1991) and subsyndromal delirium (Hwang et al., 2018), whereas female gender was related to fatigue (Hwang et al., 2014), anxiety and depression (Hu et al., 2018), and PTSD symptoms (Palgi et al., 2011). Four studies reported that lower SES was associated with depression and anxiety (Han et al., 2013; Jeong & An, 2017; Kim et al., 2017) and fatigue (Hwang et al., 2014). In addition, Liedman et al. (2001) and Hwang et al. (2014) both found that patients who had undergone total gastrectomy had more symptoms (i.e., GI symptoms and fatigue) than patients who had other types of surgery (p < 0.05). Advanced cancer stage was a risk factor that was associated with increased weight loss (Haugstvedt et al., 1991) and psychological distress (Kim et al., 2017). However, Hwang et al. (2014) and Park et al. (2015) found that advanced cancer stage was related to lower levels of fatigue. Therefore, it is difficult to draw a conclusion on predictors of a variety of symptoms. 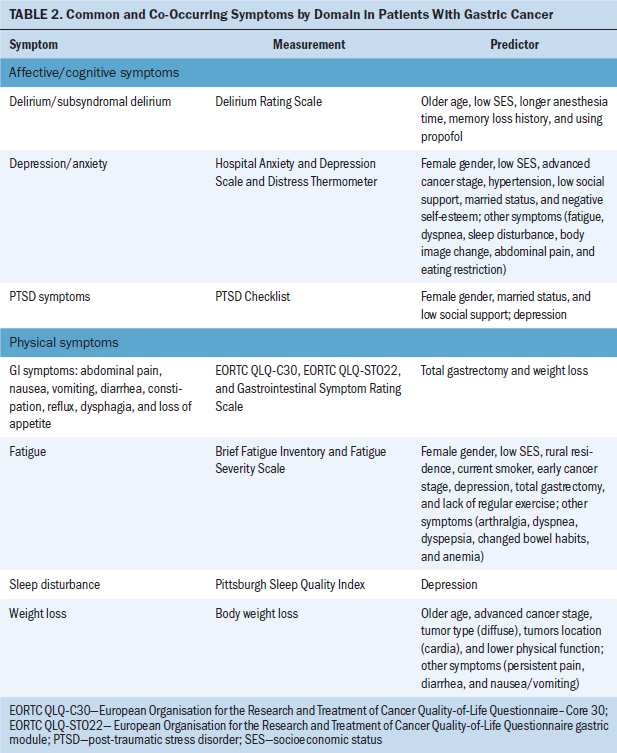
Symptom trajectories: Symptoms experienced by patients with gastric cancer were found to change over time. Four prospective longitudinal studies reported findings related to the trajectories of symptoms (Hwang et al., 2018; Nikbakhsh et al., 2016; Shim et al., 2019; Yu et al., 2016). For example, Yu et al. (2016) measured symptoms yearly for five years after surgery and found that fatigue increased significantly at year 1 and then decreased gradually (p < 0.001). This study also reported that dysphagia and eating restrictions worsened at year 1 (p < 0.001) and were at higher levels at year 5 than preoperatively (p < 0.001). In addition, Hwang et al. (2018) reported that subsyndromal delirium severity was highest on the first day after surgery and then gradually decreased in the following week (p < 0.05), whereas another study (Shim et al., 2019) found that the severity of subsyndromal delirium decreased over three days after surgery. Nikbakhsh et al. (2016) found that the severity of anxiety and depression increased from the beginning to the eighth week in the control group and decreased in the experimental group (p < 0.05). Together, these findings provide important insights into trajectories of common symptoms with short-term (days) and long-term (years) observation.
Symptom management: Interventions have been developed and tested to help patients with gastric cancer self-manage their symptoms. Four studies examined the effects of complementary and alternative approaches and pharmaceutical therapy for the management of multiple symptoms in gastric cancer, and evaluated the feasibility and efficacy of the interventions (Gunji et al., 2013; Guo & Wang, 2018; Nikbakhsh et al., 2016; Zhou et al., 2017). In a randomized controlled trial of nerve electrical stimulation treatment for chemotherapy-induced nausea and vomiting, Guo and Wang (2018) reported that the severity of nausea and vomiting decreased and loss of appetite improved significantly in 124 patients with advanced gastric cancer (p < 0.05). Similarly, another randomized controlled trial of acupuncture conducted by Zhou et al. (2017) found that the duration of nausea and abdominal pain and the frequency of vomiting and diarrhea were lower in the experimental group than in the control group (p < 0.05). In a quasiexperimental study of four-week rikkunshito (Japanese herbal therapy) administered after surgery, Gunji et al. (2013) found that the severity of abdominal pain, reflux, diarrhea, and constipation experienced by participants significantly decreased (p < 0.01). These results suggested that alternative therapies can decrease the severity and frequency of symptoms. In addition, in a randomized clinical trial of olanzapine among 30 patients with gastric cancer receiving chemotherapy (Nikbakhsh et al., 2016), the severity of anxiety and depression was lower in the experimental group compared to the control group (p < 0.05). In summary, three of four studies used different alternative therapies for GI symptom management, and one study effectively treated anxiety and depression using a pharmaceutical therapy.
Discussion
The purpose of this integrative review was to describe common and co-occurring symptoms and their symptom profiles, including measurement, predictors, trajectories, and management in patients with gastric cancer, and to address the state of symptom science in gastric cancer. The studies in this review were mainly conducted with Asian patients (21 of 25, 84%), perhaps because of higher rates of gastric cancer within this population. The latest global cancer statistics show that incidence rates of gastric cancer are highest in East Asia (particularly in Korea, Japan, and China) (Bray et al., 2018). However, the incidence of cancers of the lower stomach has been increasing among Americans aged younger than 50 years, according to a National Cancer Institute (NCI, 2018)–led study. To date, no studies have been conducted in the United States regarding symptoms in patients with gastric cancer. Therefore, more attention must be given to issues related to symptoms experienced by this population.
A number of instruments was used to assess common and co-occurring symptoms. However, no standard instrument was identified to measure multiple co-occurring symptoms in patients with gastric cancer, which set difficulties for comparison studies. The EORTC QLQ-C30 and EORTC QLQ-STO22 scales were the instruments used most frequently to measure symptoms in gastric cancer survivors. Although these two scales provide a simple and rapid assessment, they have some disadvantages. They may only allow for the evaluation of the occurrence and severity of symptoms and may not reflect other dimensions (e.g., distress, meaning), and they do not measure affective/cognitive symptoms. Several investigator-designed instruments were used, but these lacked reliability and validity indices (Park et al., 2015; Zhou et al., 2017), which may lead to inaccurate assessment of symptoms among patients with gastric cancer. In a literature review summarizing measurement tools for patient-reported outcomes in advanced gastric cancer, three additional instruments were widely used: the Functional Assessment of Cancer Therapy (FACT)-General, FACT-Gastric, and MD Anderson Symptom Inventory for GI cancer (Xu et al., 2013). In addition, two leading patient-reported outcomes (PROs) measures, the PROs version of the Common Terminology Criteria for Adverse Events and PROs Measurement Information System, developed by the National Institutes of Health, are quickly becoming the standard in patient-reported health measurement (Basch et al., 2014; Lee et al., 2020; Reeve et al., 2007). These systematically and rigorously tested instruments may provide a more comprehensive measure of multiple aspects of co-occurring symptoms in patients with gastric cancer.
The current review reported that 3–17 symptoms (median = 7) were experienced concurrently by patients with gastric cancer. Similarly, researchers have acknowledged the multiple co-occurring symptoms in patients with gastric cancer (Kawamura et al., 2014; Tey et al., 2014). In 2001, Dodd et al. first introduced the concept of symptom clusters. A symptom cluster has been defined as two or more symptoms occurring concurrently with or without sharing a common etiology (Aktas, 2013; Barsevick, 2016). It has been proposed that symptom clusters may have common underlying mechanisms that could benefit cancer symptom management (Miaskowski et al., 2017). In the past two decades, research on symptom clusters has continued to grow. To date, the authors found only one paper identifying symptom clusters in GI cancers (Han et al., 2019). However, the authors did not find any studies focused on symptom clusters in patients with gastric cancer, suggesting that symptom clusters research in this population is at an early stage of development. Future research is needed that focuses on symptom clusters to identity the phenotypes of groups of symptoms. This will enhance more efficient symptom assessment and management.
In this integrative review of 25 studies, GI symptoms (e.g., abdominal pain, dysphagia, constipation, nausea, vomiting) were among the most frequent, bothersome, and co-occurring symptoms experienced by patients with gastric cancer. This is consistent with previous findings (Cherwin, 2012; Cherwin et al., 2019) of GI symptoms being prevalent among patients with cancer. Other previous studies partially explained the etiology of this set of symptoms. For example, abdominal pain, nausea, and diarrhea are prominent issues that could be explained by bloating and abnormal movement of remnant stomach or intestine (Hejazi et al., 2010), and vomiting was because of a lower ability to store food (Kawamura et al., 2014). Increasing the need for effectively assessing and managing GI symptoms is crucial for gastric cancer survivors.
Symptom experience is a multidimensional concept with four components (Armstrong, 2003); however, very few studies have examined symptom frequency and symptom distress, and no studies have explored symptom meaning for patients with gastric cancer. These oversights are critical in the understanding of symptoms that are of particular importance, such as those based on patients’ concomitant meanings (Maguire et al., 2014). Also, the most severe or frequently occurring symptoms are not always the most distressing or meaningful to patients with cancer (Boehmke & Dickerson, 2005). These issues should be considered when systematically and comprehensively describing symptom experience in patients with gastric cancer.
Research on symptom trajectories is limited. Understanding how symptoms and symptom clusters change over time is critical to ensure appropriate symptom self-management (Dodd et al., 2001). Therefore, it is crucial to conduct longitudinal studies of symptoms in the gastric cancer population. In addition, the science behind predictors of symptoms (e.g., advanced cancer stage) reports conflicting or inconsistent results. Symptom trajectories and predictors of symptom clusters are also absent in the literature. Further investigation is needed in those areas.
Developing and testing effective symptom interventions is critical for managing co-occurring symptoms and improving quality of life for patients with cancer (Kwekkeboom, 2016). Complementary and alternative therapies have been tested for their efficacy at relieving symptoms for patients with gastric cancer. For example, herbal therapy was found to relieve the symptoms of fatigue, nausea and vomiting, pain, loss of appetite, and constipation (Xu et al., 2017). Acupuncture was also found to minimize GI symptoms after gastrectomy (Lu & Rosenthal, 2013). However, key design issues in this set of studies prevent understanding of their efficacy; these issues include small samples (19–56) and lack of well-designed randomized controlled trial designs. Therefore, further research is needed to understand the impact of alternative therapies among patients with gastric cancer who experience multiple co-occurring symptoms.
Limitations
The generalizability of the results of this literature review may be limited by several characteristics of the studies. More than half (21 of 25) of the studies are from Asian countries where gastric cancer rates are the highest globally, and the review was limited to articles written in English. Therefore, it may omit relevant articles published in other languages, particularly in Asian. There also was a very wide variability in stage of gastric cancer and stage of treatment. Studies included participants who were newly diagnosed, patients with advanced cancer, patients diagnosed with a range of earlier-stage disease (e.g., I, II), patients undergoing chemotherapy, and those who underwent gastrectomy. This variability may result in different symptoms experienced by patients with gastric cancer and, consequently, inaccurate findings on symptom profiles.
Implications for Practice and Research
The study of symptoms in patients with gastric cancer and their application to practice is vital to nursing research and practice. Three implications were identified to guide researchers and nurse clinicians in the future. First, nurses and researchers should identify common data elements (defined as variables that are operationalized and measured in identical ways across studies) for symptoms in patients with gastric cancer (Redeker et al., 2015). This would be helpful for advancing symptom science in gastric cancer and comparison across studies and populations (e.g., cancer, heart disease). Second, evidence indicates that people living with gastric cancer experience a wide range of symptoms. Healthcare providers should be directed to strengthen awareness of assessing co-occurring symptoms and symptom clusters in this population and discover potential symptoms with common etiology. Third, clinicians and researchers should partner to develop innovative interventions to support self-management of symptoms, including targeting the symptom clusters instead of individual symptoms. Further exploration of symptom clusters will provide a foundation for developing future interventions for efficient, effective symptom management in patients with gastric cancer. 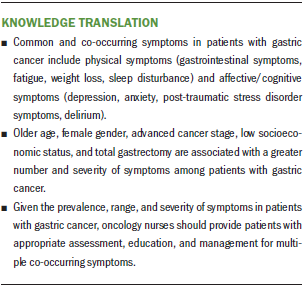
Conclusion
The authors reviewed relevant studies on symptom experience, measurement, predictors, trajectories, and management in patients with gastric cancer. This area of science remains in its infancy because robust evidence related to these symptoms is not available. Symptom predictors and trajectories have yet to be fully studied and described in patients with gastric cancer. Identification of symptom clusters may help to determine how symptoms are related to one another and how they influence patients’ outcomes. This emphasizes the need for further research to establish the science. Interventions targeted to symptom clusters may help to improve the efficacy of symptom management in patients with gastric cancer.
About the Author(s)
Yufen Lin, MSN, RN, is a PhD student in the School of Nursing, Sharron L. Docherty, PhD, PNP, FAAN, is an associate professor in the School of Nursing, Laura S. Porter, PhD, is a professor in the School of Medicine, Department of Psychiatry and Behavioral Sciences, and Donald E. Bailey Jr., PhD, RN, FAAN, is an associate professor in the School of Nursing, all at Duke University in Durham, NC. No financial relationships to disclose. All authors contributed to the conceptualization and design and manuscript preparation. Lin and Bailey completed the data collection, evaluation, and analysis. Lin can be reached at yufen.lin@duke.edu, with copy to ONFEditor@ons.org. (Submitted May 2019. Accepted August 7, 2019.)
References
Aktas, A. (2013). Cancer symptom clusters: Current concepts and controversies. Current Opinion in Supportive and Palliative Care, 7(1), 38–44. https://doi.org/10.1097/SPC.0b013e32835def5b
Albrecht, T.A. (2014). Physiologic and psychological symptoms experienced by adults with acute leukemia: An integrative literature review. Oncology Nursing Forum, 41(3), 286–295. https://doi.org/10.1188/14.ONF.286-295
Anderson, I.D., & MacIntyre, I.M.C. (1995). Symptomatic outcome following resection of gastric cancer. Surgical Oncology, 4(1), 35–40. https://doi.org/10.1016/s0960-7404(10)80029-5
Armstrong, T.S. (2003). Symptoms experience: A concept analysis. Oncology Nursing Forum, 30(4), 601–606. https://doi.org/10.1188/03.ONF.601-606
Axon, A. (2006). Symptoms and diagnosis of gastric cancer at early curable stage. Best Practice and Research Clinical Gastroenterology, 20(4), 697–708. https://doi.org/10.1016/j.bpg.2006.03.015
Barad, A.K., Mandal, S.K., Harsha, H.S., Sharma, B.M., & Singh, T.S. (2014). Gastric cancer—A clinicopathological study in a tertiary care centre of North-eastern India. Journal of Gastrointestinal Oncology, 5(2), 142–147. https://doi.org/10.3978/j.issn.2078-6891.2014.003
Barsevick, A. (2016). Defining the symptom cluster: How far have we come? Seminars in Oncology Nursing, 32(4), 334–350. https://doi.org/10.1016/j.soncn.2016.08.001
Basch, E., Reeve, B.B., Mitchell, S.A., Clauser, S.B., Minasian, L.M., Dueck, A.C., . . . Schrag, D. (2014). Development of the National Cancer Institute’s Patient-Reported Outcomes version of the Common Terminology Criteria for Adverse Events (PRO-CTACE). Journal of the National Cancer Institute, 106(9), dju244. https://doi.org/10.1093/jnci/dju244
Boehmke, M.M., & Dickerson, S.S. (2005). Symptom, symptom experiences, and symptom distress encountered by women with breast cancer undergoing current treatment modalities. Cancer Nursing, 28(5), 382–389. https://doi.org/10.1097/00002820-200509000-00008
Bower, J.E. (2008). Behavioral symptoms in patients with breast cancer and survivors. Journal of Clinical Oncology, 26(5), 768–777. https://doi.org/10.1200/jco.2007.14.3248
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R.L., Torre, L.A., & Jemal, A. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: A Cancer Journal for Clinicians, 68(6), 394–424. https://doi.org/10.3322/caac.21492
Burrell, S.A., Yeo, T.P., Smeltzer, S.C., Leiby, B.E., Lavu, H., Kennedy, E.P., & Yeo, C.J. (2018a). Symptom clusters in patients with pancreatic cancer undergoing surgical resection: Part I. Oncology Nursing Forum, 45(4), E36–E52. https://doi.org/10.1188/18.ONF.e36-e52
Burrell, S.A., Yeo, T.P., Smeltzer, S.C., Leiby, B.E., Lavu, H., Kennedy, E.P., & Yeo, C.J. (2018b). Symptom clusters in patients with pancreatic cancer undergoing surgical resection: Part II. Oncology Nursing Forum, 45(4), E53–E66. https://doi.org/10.1188/18.ONF.e53-e66
Cherwin, C., Nakad, L., & Albashayreh, A. (2019). Systematic review of nonpharmacologic approaches for the management of gastrointestinal symptoms. Oncology Nursing Forum, 46(1), E1–E21. https://doi.org/10.1188/19.ONF.e1-e21
Cherwin, C.H. (2012). Gastrointestinal symptom representation in cancer symptom clusters: A synthesis of the literature. Oncology Nursing Forum, 39(2), 157–165. https://doi.org/10.1188/12.ONF.157-165
Cho, M.H. (2004). Sleep disturbances in gastric cancer patients and their family caregivers [Doctoral dissertation, University of California, San Francisco]. ProQuest. https://search.proquest.com/openview/639aff7f4498974c6a16713850abf7b1/1…
Climent, M., Munarriz, M., Blazeby, J.M., Dorcaratto, D., Ramón, J.M., Carrera, M.J., . . . Pera, M. (2017). Weight loss and quality of life in patients surviving 2 years after gastric cancer resection. European Journal of Surgical Oncology, 43(7), 1337–1343. https://doi.org/10.1016/j.ejso.2017.01.239
Cooley, M.E. (2000). Symptoms in adults with lung cancer: A systematic research review. Journal of Pain and Symptom Management, 19(2), 137–153. https://doi.org/10.1016/S0885-3924(99)00150-5
Dodd, M.J., Miaskowski, C., & Paul, S.M. (2001). Symptom clusters and their effect on the functional status of patients with cancer. Oncology Nursing Forum, 28(3), 465–470.
Garrard, J. (2014). Health sciences literature review made easy: The matrix method (4th ed.). Jones and Bartlett Learning.
Gunji, S., Ueda, S., Yoshida, M., Kanai, M., Terajima, H., & Takabayashi, A. (2013). Effects of rikkunshito, a kampo medicine, on quality of life after proximal gastrectomy. Journal of Surgical Research, 185(2), 575–580. https://doi.org/10.1016/j.jss.2013.06.010
Guo, W.C., & Wang, F. (2018). Effect of nerve electrical stimulation for treating chemotherapy-induced nausea and vomiting in patients with advanced gastric cancer: A randomized controlled trial. Medicine, 97(51), e13620. https://doi.org/10.1097/md.0000000000013620
Han, C.J., Reding, K., Cooper, B.A., Paul, S.M., Conley, Y.P., Hammer, M., . . . Miaskowski, C. (2019). Symptom clusters in patients with gastrointestinal cancers using different dimensions of the symptom experience. Journal of Pain and Symptom Management, 58(2), 224–234. https://doi.org/10.1016/j.jpainsymman.2019.04.035
Han, K.H., Hwang, I.C., Kim, S., Bae, J.M., Kim, Y.W., Ryu, K.W., . . . Yun, Y.H. (2013). Factors associated with depression in disease-free stomach cancer survivors. Journal of Pain and Symptom Management, 46(4), 511–522. https://doi.org/10.1016/j.jpainsymman.2012.10.234
Haugstvedt, T.K., Viste, A., Eide, G.E., & Søreide, O. (1991). Factors related to and consequences of weight loss in patients with stomach cancer. The Norwegian multicenter experience. Cancer, 67(3), 722–729. https://doi.org/10.1002/1097-0142(19910201)67:3<722::aid-cncr2820670332…
Hejazi, R.A., Patil, H., & McCallum, R.W. (2010). Dumping syndrome: Establishing criteria for diagnosis and identifying new etiologies. Digestive Diseases and Sciences, 55(1), 117–123. https://doi.org/10.1007/s10620-009-0939-5
Hirshkowitz, M., Whiton, K., Albert, S.M., Alessi, C., Bruni, O., DonCarlos, L., . . . Adams Hillard, P.J. (2015). National Sleep Foundation’s sleep time duration recommendations: Methodology and results summary. Sleep Health, 1(1), 40–43. https://doi.org/10.1016/j.sleh.2014.12.010
Hockenberry, M.J., Hooke, M.C., Rodgers, C., Taylor, O., Koerner, K.M., Mitby, P., . . . Pan, W. (2017). Symptom trajectories in children receiving treatment for leukemia: A latent class growth analysis with multitrajectory modeling. Journal of Pain and Symptom Management, 54(1), 1–8. https://doi.org/10.1016/j.jpainsymman.2017.03.002
Hong, J., Wei, Z., & Wang, W. (2015). Preoperative psychological distress, coping and quality of life in Chinese patients with newly diagnosed gastric cancer. Journal of Clinical Nursing, 24(17–18), 2439–2447. https://doi.org/10.1111/jocn.12816
Hu, L.Y., Liu, C.J., Yeh, C.M., Lu, T., Hu, Y.W., Chen, T.J., . . . Chang, C.H. (2018). Depressive disorders among patients with gastric cancer in Taiwan: A nationwide population-based study. BMC Psychiatry, 18(1), 272–279. https://doi.org/10.1186/s12888-018-1859-8
Hwang, H., Lee, K.M., Son, K.L., Jung, D., Kim, W.H., Lee, J.Y., . . . Hahm, B.J. (2018). Incidence and risk factors of subsyndromal delirium after curative resection of gastric cancer. BMC Cancer, 18(1), 765–774. https://doi.org/10.1186/s12885-018-4681-2
Hwang, I.C., Yun, Y.H., Kim, Y.W., Ryu, K.W., Kim, Y.A., Kim, S., . . . Sohn, T.S. (2014). Factors related to clinically relevant fatigue in disease-free stomach cancer survivors and expectation-outcome consistency. Supportive Care in Cancer, 22, 1453–1460. https://doi.org/10.1007/s00520-013-2110-2
Jaffee, E.M., Van Dang, C., Agus, D.B., Alexander, B.M., Anderson, K.C., Ashworth, A., . . . Yung, A. (2017). Future cancer research priorities in the USA: A Lancet Oncology commission. Lancet Oncology, 18(11), e653–e706. https://doi.org/10.1016/S1470-2045(17)30698-8
Jeong, A., & An, J.Y. (2017). The moderating role of social support on depression and anxiety for gastric cancer patients and their family caregivers. PLOS ONE, 12(12), e0189808. https://doi.org/10.1371/journal.pone.0189808
Karimi, P., Islami, F., Anandasabapathy, S., Freedman, N.D., & Kamangar, F. (2014). Gastric cancer: Descriptive epidemiology, risk factors, screening, and prevention. Cancer Epidemiology, Biomarkers and Prevention, 23(5), 700–713. https://doi.org/10.1158/1055-9965.Epi-13-1057
Kawamura, H., Takahashi, N., Homma, S., Minagawa, N., Shibasaki, S., Takahashi, M., & Taketomi, A. (2014). Assessment of postoperative symptoms after laparoscopy-assisted distal gastrectomy for stage I gastric cancer. International Surgery, 99(5), 645–649. https://doi.org/10.9738/intsurg-d-13-00111.1
Kim, G.M., Kim, S.J., Song, S.K., Kim, H.R., Kang, B.D., Noh, S.H., . . . Rha, S.Y. (2017). Prevalence and prognostic implications of psychological distress in patients with gastric cancer. BMC Cancer, 17(1), 283. https://doi.org/10.1186/s12885-017-3260-2
Kim, S.M., Cho, J., Kang, D., Oh, S.J., Kim, A.R., Sohn, T.S., . . . Kim, S. (2016). A randomized controlled trial of vagus nerve-preserving distal gastrectomy versus conventional distal gastrectomy for postoperative quality of life in early stage gastric cancer patients. Annals of Surgery, 263(6), 1079–1084. https://doi.org/10.1097/sla.0000000000001565
Konishi, H., Nakada, K., Kawamura, M., Iwasaki, T., Murakami, K., Mitsumori, N., & Yanaga, K. (2016). Impaired gastrointestinal function affects symptoms and alimentary status in patients after gastrectomy. World Journal of Surgery, 40(11), 2713–2718. https://doi.org/10.1007/s00268-016-3613-z
Kwekkeboom, K.L. (2016). Cancer symptom cluster management. Seminars in Oncology Nursing, 32(4), 373–382. https://doi.org/10.1016/j.soncn.2016.08.004
Kwekkeboom, K.L., Cherwin, C.H., Lee, J.W., & Wanta, B. (2010). Mind-body treatments for the pain-fatigue-sleep disturbance symptom cluster in persons with cancer. Journal of Pain and Symptom Management, 39(1), 126–138.
Lee, L., Ross, A., Griffith, K., Jensen, R.E., & Wallen, G.R. (2020). Symptom clusters in breast cancer survivors: A latent class profile analysis. Oncology Nursing Forum, 47(1), 89–100. https://doi.org/10.1188/20.ONF.89-100
Lee, S.S., Chung, H.Y., Kwon, O., & Yu, W. (2016). Long-term quality of life after distal subtotal and total gastrectomy: Symptom- and behavior-oriented consequences. Annals of Surgery, 263(4), 738–744. https://doi.org/10.1097/sla.0000000000001481
Liedman, B., Svedlund, J., Sullivan, M., Larsson, L., & Lundell, L. (2001). Symptom control may improve food intake, body composition, and aspects of quality of life after gastrectomy in cancer patients. Digestive Diseases and Sciences, 46(12), 2673–2680. https://doi.org/10.1023/a:1012719211349
Lu, W., & Rosenthal, D.S. (2013). Acupuncture for cancer pain and related symptoms. Current Pain and Headache Reports, 17(3), 321. https://doi.org/10.1007/s11916-013-0321-3
Maconi, G., Kurihara, H., Panizzo, V., Russo, A., Cristaldi, M., Marrelli, D., . . . Taschieri, A.M. (2003). Gastric cancer in young patients with no alarm symptoms: Focus on delay in diagnosis, stage of neoplasm and survival. Scandinavian Journal of Gastroenterology, 38(12), 1249–1255.
Maeda, T., & Munakata, T. (2008). The effect of appropriate eating habits, depressive state, and social support on postoperative symptom experience among Japanese postgastrectomy patients. Gastroenterology Nursing, 31(6), 423–429. https://doi.org/10.1097/SGA.0b013e31818ff081
Maeda, T., Onuoha, F.N., & Munakata, T. (2006). The effect of postoperative symptom experience, and personality and psychosocial factors on depression among postgastrectomy patients in Japan. Gastroenterology Nursing, 29(6), 437–444. https://doi.org/10.1097/00001610-200611000-00002
Maguire, R., Stoddart, K., Flowers, P., McPhelim, J., & Kearney, N. (2014). An interpretative phenomenological analysis of the lived experience of multiple concurrent symptoms in patients with lung cancer: A contribution to the study of symptom clusters. European Journal of Oncology Nursing, 18(3), 310–315. https://doi.org/10.1016/j.ejon.2014.02.004
Melnyk, B.M., & Fineout-Overholt, E. (2011). Evidence-based practice in nursing and healthcare: A guide to best practice (2nd ed.). Wolters Kluwer Health.
Miaskowski, C., Barsevick, A., Berger, A., Casagrande, R., Grady, P.A., Jacobsen, P., . . . Marden, S. (2017). Advancing symptom science through symptom cluster research: Expert panel proceedings and recommendations. Journal of the National Cancer Institute, 109(4), djw253. https://doi.org/10.1093/jnci/djw253
Miaskowski, C., Dodd, M., & Lee, K. (2004). Symptom clusters: The new frontier in symptom management research. JNCI Monographs, 2004(32), 17–21. https://doi.org/10.1093/jncimonographs/lgh023
Mine, S., Sano, T., Tsutsumi, K., Murakami, Y., Ehara, K., Saka, M., . . . Katai, H. (2010). Large-scale investigation into dumping syndrome after gastrectomy for gastric cancer. Journal of the American College of Surgeons, 211(5), 628–636. https://doi.org/10.1016/j.jamcollsurg.2010.07.003
National Cancer Institute. (2018, January 18). Incidence of cancers of the lower stomach increasing among younger Americans. Cancer Currents Blog. https://www.cancer.gov/news-events/cancer-currents-blog/2018/lower-stom…
National Institute of Nursing Research. (2016). The NINR Strategic Plan: Advancing science, improving lives: A vision for Nursing Science. https://www.ninr.nih.gov/sites/files/docs/NINR_StratPlan2016_reduced.pdf
National Institute of Nursing Research. (2019). Symptom science center: A resource for precision health. https://www.ninr.nih.gov/newsandinformation/events/sscevent
National Institutes of Health State-of-the-Science Panel. (2004). National Institutes of Health State-of-the-Science Conference statement: Symptom management in cancer: Pain, depression, and fatigue, July 15–17, 2002. Journal of the National Cancer Institute Monographs, 2004(32), 9–16. https://doi.org/10.1093/jncimonographs/djg014
Nikbakhsh, N., Sadeghi, M.V., Ramzani, E., Moudi, S., Bijani, A., Yousefi, R., . . . Gholinia, H. (2016). Efficacy of olanzapine in symptom relief and quality of life in gastric cancer patients receiving chemotherapy. Journal of Research in Medical Sciences, 21, 88. https://doi.org/10.4103/1735-1995.192504
Oh, H.J., Choi, M.G., Park, J.M., Song, K.Y., & Yoo, H.M. (2018). Acid secretion and its relationship to esophageal reflux symptom in patients with subtotal gastrectomy. Digestive Diseases and Sciences, 63(3), 703–712. https://doi.org/10.1007/s10620-018-4923-9
Palgi, Y., Shrira, A., Haber, Y., Wolf, J.J., Goldray, O., Shacham-Shmueli, E., & Ben-Ezra, M. (2011). Comorbidity of posttraumatic stress symptoms and depressive symptoms among gastric cancer patients. European Journal of Oncology Nursing, 15(5), 454–458. https://doi.org/10.1016/j.ejon.2010.11.011
Park, W., Lee, J.K., Kim, C.R., & Shin, J.Y. (2015). Factors associated with fatigue in Korean gastric cancer survivors. Korean Journal of Family Medicine, 36(6), 328–334. https://doi.org/10.4082/kjfm.2015.36.6.328
Pettersson, G., Berterö, C., Unosson, M., & Börjeson, S. (2014). Symptom prevalence, frequency, severity, and distress during chemotherapy for patients with colorectal cancer. Supportive Care in Cancer, 22(5), 1171–1179. https://doi.org/10.1007/s00520-013-2069-z
Rausei, S., Mangano, A., Galli, F., Rovera, F., Boni, L., Dionigi, G., & Dionigi, R. (2013). Quality of life after gastrectomy for cancer evaluated via the EORTC QLQ-C30 and QLQ-STO22 questionnaires: Surgical considerations from the analysis of 103 patients. International Journal of Surgery, 11(S1), s104–s109. https://doi.org/10.1016/s1743-9191(13)60028-x
Redeker, N.S., Anderson, R., Bakken, S., Corwin, E., Docherty, S., Dorsey, S.G., . . . Grady, P. (2015). Advancing symptom science through use of common data elements. Journal of Nursing Scholarship, 47(5), 379–388. https://doi.org/10.1111/jnu.12155
Reeve, B.B., Hays, R.D., Bjorner, J.B., Cook, K.F., Crane, P.K., Teresi, J.A., . . . Cella, D. (2007). Psychometric evaluation and calibration of health-related quality of life item banks: Plans for the patient-reported outcomes measurement information system (PROMIS). Medical Care, 45(5), s22–s31. https://doi.org/10.1097/01.mlr.0000250483.85507.04
Shim, E.J., Noh, H.L., Lee, K.M., Hwang, H., Son, K.L., Jung, D., . . . Hahm, B.J. (2019). Trajectory of severity of postoperative delirium symptoms and its prospective association with cognitive function in patients with gastric cancer: Results from a prospective observational study. Supportive Care in Cancer, 27(8), 2999–3006. https://doi.org/10.1007/s00520-018-4604-4
Siegel, R.L., Miller, K.D., & Jemal, A. (2018). Cancer statistics, 2018. CA: A Cancer Journal for Clinicians, 68(1), 7–30. https://doi.org/10.3322/caac.21442
Talcott, J.A., Manola, J., Clark, J.A., Kaplan, I., Beard, C.J., Mitchell, S.P., . . . D’Amico, A.V. (2003). Time course and predictors of symptoms after primary prostate cancer therapy. Journal of Clinical Oncology, 21(21), 3979–3986. https://doi.org/10.1200/jco.2003.01.199
Tchen, N., Juffs, H.G., Downie, F.P., Yi, Q.L., Hu, H., Chemerynsky, I., . . . Tannock, I.F. (2003). Cognitive function, fatigue, and menopausal symptoms in women receiving adjuvant chemotherapy for breast cancer. Journal of Clinical Oncology, 21(22), 4175–4183. https://doi.org/10.1200/JCO.2003.01.119
Tey, J., Choo, B.A., Leong, C.N., Loy, E.Y., Wong, L.C., Lim, K., . . . Koh, W.Y. (2014). Clinical outcome of palliative radiotherapy for locally advanced symptomatic gastric cancer in the modern era. Medicine, 93(22), e118. https://doi.org/10.1097/MD.0000000000000118
Venerito, M., Vasapolli, R., Rokkas, T., & Malfertheiner, P. (2018). Gastric cancer: Epidemiology, prevention, and therapy. Helicobacter, 23(S1), e12518. https://doi.org/10.1111/hel.12518
Wong, M.L., Cooper, B.A., Paul, S.M., Levine, J.D., Conley, Y.P., Wright, F., . . . Miaskowski, C. (2017). Differences in symptom clusters identified using ratings of symptom occurrence vs. severity in lung cancer patients receiving chemotherapy. Journal of Pain Symptom Management, 54(2), 194–203. https://doi.org/10.1016/j.jpainsymman.2017.04.005
Xu, J., Evans, T.R.J., Coon, C., Copley-Merriman, K., & Su, Y. (2013). Measuring patient-reported outcomes in advanced gastric cancer. ecancermedicalscience, 7, 351. https://doi.org/10.3332/ecancer.2013.351
Xu, Y., Mao, J.J., Sun, L., Yang, L., Li, J., Hao, Y., . . . Yang, Y. (2017). Association between use of traditional Chinese medicine herbal therapy and survival outcomes in patients with stage II and III colorectal cancer: A multicenter prospective cohort study. JNCI Monographs, 2017(52), lgx015. https://doi.org/10.1093/jncimonographs/lgx015
Yu, W., Park, K.B., Chung, H.Y., Kwon, O.K., & Lee, S.S. (2016). Chronological changes of quality of life in long-term survivors after gastrectomy for gastric cancer. Cancer Research and Treatment, 48(3), 1030–1036. https://doi.org/10.4143/crt.2015.398
Zhou, J., Fang, L., Wu, W.Y., He, F., Zhang, X.L., Zhou, X., & Xiong, Z.J. (2017). The effect of acupuncture on chemotherapy-associated gastrointestinal symptoms in gastric cancer. Current Oncology, 24(1), e1–e5. https://doi.org/10.3747/co.24.3296




