Causes and Consequences of Chemotherapy Delays in Ambulatory Oncology Practices: A Multisite Qualitative Study
Purpose: In oncology, chemotherapy treatment delays potentially jeopardize patient safety and impede progress toward disease remission. The purpose of this study was to examine the causes and consequences of chemotherapy treatment delays and possible solutions to improve quality of care.
Participants & Setting: The current authors selected a purposive sample of eight ambulatory oncology practices for ethnographic site visits, which lasted five days each.
Methodologic Approach: The authors conducted 290 observation hours, including clinician shadowing, and 46 semistructured interviews with clinicians (oncology nurses, physicians, and advanced practice providers). Deductive and inductive thematic analysis was performed on all data.
Findings: The authors identified four primary themes from the analysis that affect delays: discrepancies in care plans and missing orders, uncommunicated day-of-treatment order changes, orders not signed in advance by physicians, and laboratory testing processes.
Implications for Nursing: Future investigations should examine nurses’ communication practices in the context of timely chemotherapy administration because communication and documentation technologies within healthcare settings continuously evolve.
Jump to a section
Timely access to care in ambulatory oncology settings is a priority. More than 80% of all healthcare encounters for chemotherapy occur at ambulatory oncology practices (National Center for Health, 2020). Concerns from the public and private sectors about the unsatisfactory timeliness of health interventions informed one of the six key domains of the Institute of Medicine (2001) framework for healthcare quality. Chemotherapy treatment delays or wait times impede progress toward optimal health outcomes and place patients at risk for unfavorable events, such as missed medication administration, suboptimal disease control, or death (Joint Commission, 2015).
Previous work has aimed to understand to what extent patients, clinicians, and organizational factors of the health system influence delays in health care. For example, in primary care settings, staffing and scheduling policies of the organization may facilitate reduced wait times (Ansell et al., 2017). Previous research about delays within clinical oncology has emphasized that the time from diagnosis to treatment is a significant indicator of quality (Honein-AbouHaidar et al., 2017). However, patient-centered issues in the oncology continuum extend beyond diagnosis and should be emphasized during treatment and survivorship. The current authors define delays as the prolonged and avoidable periods of time that it takes for patients to receive chemotherapy during their scheduled appointment day and time.
As the cancer care landscape in the United States is changing rapidly, with a flurry of practice closures and consolidations reported (Community Oncology Alliance, 2018), research to identify patterns, correlates, and consequences of delays across diverse cancer settings is needed. The purpose of this study was to examine the phenomena of chemotherapy treatment delays and generate possible solutions to improve quality of care.
Conceptual Framework
The conceptual framework guiding this inquiry (see Figure 1) was derived from a sociotechnical theoretical framework incorporating concepts from communication, information technology, and organizational studies (Bostrom & Heinen, 1977; Singh & Sittig, 2016; Zheng, Ciemins, et al., 2015; Zheng, Hanauer, et al., 2015). The framework informed the qualitative data collection protocol and interview guides described in this article. Briefly, the model posits that communication and breakdowns in communication are potentially driving forces in quality-of-care deficits observed in ambulatory oncology settings. Solutions may include human resources, technology changes, or efforts to shift organizational culture. 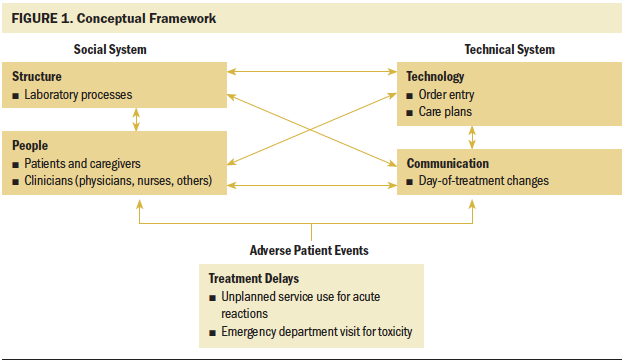
Given the alignment between the study’s conceptual model and the methods used in the current inquiry, the study team was able to view delays in chemotherapy treatment from diverse perspectives, including interactions between patients and clinicians. Therefore, the team could gain a better understanding of the nuances in the causes and consequences of treatment delays and highlight future opportunities for feasible solutions.
Methods
Participants and Setting
This study is part of a sequential, mixed-methods project examining clinician communication and communication technologies in ambulatory oncology practices. Potential participants were from 48 ambulatory oncology practices that are in a statewide, practice-based quality improvement program focused on cancer care delivery in Michigan. The project had three phases: patient and clinician surveys; in-depth observation, shadowing, and interviews; and focus groups.
In the first phase of the study in 2017, 29 of the 48 practices participated in survey collection over a six-week period via anonymous paper surveys, which were distributed by lead study staff at each site (Patel et al., 2019). The survey response rate among the 29 practices was 68% (n = 297). Clinician surveys elicited information about communication satisfaction, practice environments, and communication technology at the ambulatory oncology practices.
The clinicians included RNs, physicians, nurse practitioners, and physician assistants who managed patient care before, during, and after chemotherapy infusions. The authors selected a purposive sample of 8 of the 29 practices to visit during the subsequent qualitative phase of the study. Based on the 297 clinician surveys received, the authors selected practices based on variation in communication and technology ratings. Specifically, the authors constructed practice-level means from clinician ratings of the efficacy and usage of their electronic health record technology and the quality of clinician-to-clinician communication (Patel et al., 2019). The authors then arrayed the 29 practices into one of four quadrants: (a) high technology use, favorable clinician communication; (b) low technology use, favorable clinician communication; (c) high technology use, unfavorable clinician communication; and (d) low technology use, unfavorable clinician communication. Two practices were selected from each of the four quadrants.
The eight ambulatory practices were located in different geographic areas of the state and ranged in size from 7 to 34 infusion chairs. Table 1 provides additional characteristics about the eight practices, including the average daily staffing of clinicians and any affiliated health networks. This project was approved by the institutional review board at the University of Michigan. To protect the confidentiality of the sites and participants, identifying details have been removed from this article. 
In the second phase of this study, the authors conducted multimethod qualitative research at the eight selected ambulatory oncology practices. In this phase, researchers spent five days embedded within each practice conducting observation, shadowing, and interviews with clinicians. M.L., an anthropologist, was at all eight sites and was joined by one to two research team members at four sites, totaling 290 hours of data collection. Visiting sites allowed the authors to gain a holistic, nuanced understanding of the chemotherapy delivery processes at each practice.
Observation and Shadowing
The authors conducted observation in infusion areas, examination rooms, and clinician offices to understand the day-to-day chemotherapy delivery processes and organizational work structures of each practice. This method allowed for capture of verbal and nonverbal communication processes and identification of clinician task behaviors. The authors could compare what they saw with what clinicians told them and were able to bring up any site-specific discussion points during succeeding clinician interviews.
The data collection team also shadowed individual clinicians for several hours at a time to understand their role responsibilities, workflow, and communication practices. For example, the authors shadowed physicians as they saw patients and entered orders and progress notes and shadowed nurses as they administered treatments (infusion nurses), managed patients (clinic nurses), and conducted patient educational sessions for patients before their first chemotherapy treatment. Patient perspectives were gleaned from observations of patient and clinician interactions. During observation and shadowing, the authors wrote field notes, which were typed into more detailed narrative accounts used during data analysis after each day of data collection.
Interviews
After several days of observation, the authors conducted interviews, which were audio recorded, with clinicians. Although most interviews (n = 40) were one-on-one with individual clinicians, the authors conducted two small group interviews with nurses after their shifts to accommodate their schedules. In total, 46 clinicians were interviewed (see Table 2). Interview questions were designed to elicit clinicians’ perspectives of communication processes and barriers and facilitators to providing patient care. During interviews, the authors asked about specific challenges observed at that practice and asked for possible solutions to help improve patient care delivery. 
Focus Groups
After the eight site visits, focus groups were held with clinicians and clinical leaders from practices that participate in the same statewide consortium. By speaking with clinicians and clinical leaders who work at six additional practices, the authors were able to see if the challenges identified, such as delays of care, resonated beyond the sample of the eight visited sites. Four focus groups were held at two practices across the state, and two focus groups were held at a scheduled professional meeting. Participants included 7 prescribers (6 physicians, 1 nurse practitioner), 18 nurses (12 infusion, 3 triage, 3 clinic), and 8 practice administrators.
Data Analysis
The authors began data analysis after each site visit to determine if they needed to adjust the methods for data collection at the subsequent sites. Audio recordings from the interviews were transcribed verbatim, removing any identifying information. Deductive and inductive content analysis was used to analyze the data. The authors began with deductive analysis, having two research team members independently read through each transcript, listing key points organized by domains in the interview guide. These summaries were then combined and entered into a matrix spreadsheet organized by site and clinician role. The team discussed themes from each site that influenced care delivery and refined the interview questions to focus on any important emergent factors at future sites. The team met frequently and compared similarities and differences across sites as dominant patterns and themes emerged. The same process of analysis was used for the focus groups conducted after the site visits.
Upon completion of the deductive analysis, the authors re-analyzed the data using an inductive approach to identify the nuances within the text. Multiple team members reread interview transcripts and field notes to refine the dominant themes to create a codebook. Researchers used the finalized codebook to code the data from field notes, interviews, and focus groups using the qualitative data management software ATLAS.ti.
Findings
Through analysis, the authors identified four themes that led to chemotherapy treatment delays throughout the practices in the current study: (a) discrepancies in care plans and missing orders for uncoupled visits, (b) undocumented and uncommunicated day-of-treatment order changes, (c) orders not signed in advance by physicians, and (d) laboratory testing processes. The delays identified occurred at various stages within care processes and stemmed from barriers within organizational structure, communication and coordination, and communication technologies. Figure 2 illustrates where the delays occur within the flow of patient care and information transfer. In addition to describing the challenges sites experienced with treatment delays, the authors also identified strategies and policies sites had in place and suggestions from clinicians to reduce delays. 
Discrepancies in Care Plan and Missing Orders for Uncoupled Visits
When patients came for a chemotherapy infusion on days they did not have an appointment to see their physician (i.e., an uncoupled visit), delays occurred if there were discrepancies in the care plan and the prescribing physician was not on site to clarify the intended treatment. Often, on-call physicians were hesitant to make treatment decisions for unfamiliar patients when orders, care plans, and progress notes were unclear or undocumented:
A lot of times [errors occur] because . . . the provider has dictated something different . . . discrepancies in what they’ve communicated is the plan, and if they’re not there to ask, we’ve not known what to do. . . . [We’ve] sent patients home . . . that drove a couple of hours, and the on-call provider wasn’t willing to make the call because it was not clear what his primary doctor wanted. (Infusion nurse)
Care plan discrepancies between the original and on-call physician resulted in patients having their treatments canceled on the same day as their appointments, which led to treatment delays of days or weeks in some circumstances. This was a notable problem at one site, where delays were compounded by the long distances patients traveled to receive treatment.
Practices had various strategies in place to identify and address potential delay-causing issues that may arise for patients. Preparation usually entailed looking at the next day’s scheduled patients to assess laboratory values and any anticipated problems, to ensure necessary drugs were in stock, and to rectify any errors or discrepancies from orders that were not signed in advance. For practices that experienced frequent delays because of order discrepancies and poor coordination of care, such processes and responsibilities outlined previously were not clearly assigned. In some instances, infusion nurses felt it was the responsibility of office or clinic nurses to prepare for patient infusion visits. Despite efforts by an infusion lead nurse to check and clean up orders ahead of scheduled infusion visits, unclear role responsibilities and poor coordination between infusion and clinic led to details falling through the cracks, resulting in delays.
In contrast, practices that were more successful with preventing these types of delays had standardized processes in place and preparation responsibilities that were clearly assigned to specific clinician roles. For example, in certain practices, it is the clinic nurse’s responsibility to ensure that everything is prepared for patients to receive chemotherapy at their appointment time. In other practices, it is the infusion nurses who are responsible for preparing orders and ensuring that patients will be ready for their infusion visit the next day. Daily huddles were used at several sites to improve coordination between clinic and infusion nurses and to help ensure that patient preparation was handled properly and that any issues were addressed.
Undocumented and Uncommunicated Day-of-Treatment Order Changes
When patients see their prescriber on the same day as their scheduled infusion appointments (i.e., coupled visits), the prescriber may make a change to their treatment plan because of aberrant laboratory results or physical assessment findings. Although physicians may adjust the orders in the electronic health record, they sometimes move on to the next patient without updating the progress notes or communicating the last-minute order changes to infusion nurses. In these situations, physicians struggle to balance their time between seeing patients and their charting responsibilities.
When prescribers do not communicate order changes, either through informing infusion nurses or making progress notes, infusion nurses must then verify changes with prescribers prior to administering treatment. Getting clarification from the physician can take as long as 60 minutes or more, causing a delay in chemotherapy treatment. Infusion nurses commented that every time they had to go find a physician to verify an order, it took them away from the infusion floor and caring for their patients:
I know that [prescribers are] very busy . . . but if they would just stop and take that moment to communicate with nurses that there’s been a dose change or that I am holding a chemo for this reason . . . then we wouldn’t have to be tracking them down and waiting.
I think another challenge is sometimes trying to figure out what’s on the doctor’s mind. Because the patient will come in and tell us, “Oh, he said this.” They didn’t write that in their note. . . . Their progress note has not been typed up yet, or some of the progress notes are just poor and don’t give you an idea of what their plan is. So, then we still have to go back [to clarify].
In addition, when patients and nurses have a divergent understanding of the treatment plan, it can erode the patients’ trust and confidence in the level of care they are receiving, forcing infusion nurses to mask their confusion while seeking clarification from a physician:
There seems to be some kind of communication breakdown. . . . The patient knows more sometimes than we do. And then . . . you don’t want to let them know that you have no idea what’s going on.
Orders Not Signed in Advance by Physicians
Some physicians will not sign orders in advance and insist on seeing their patients the day of treatment before they sign. This causes delays in treatment when physicians move on to see their next patient without signing the previous patient’s order; meanwhile, the patient has gone to infusion and has to wait while nurses track down the physicians to sign the orders:
One of the biggest challenges we have as infusion nurses . . . is having our orders preapproved by the physicians in the electronic record so that we can go ahead with treatment. They’re supposed to have them approved before the patient’s scheduled. That doesn’t always happen, and we find ourselves at the last minute standing in front of a patient saying, “I’ll be right back.”
In response to order-signing delays, some infusion nurses use workarounds to counter the negative effects on their workflow and help improve patient care and wait times. These workarounds include fully staffing the infusion center when certain oncologists see patients, continually messaging the office reminders to sign orders, and keeping patients’ charts open in the electronic health record, so the physician has to call the nurse to access the chart and, therefore, serves as a notification for beginning treatment.
Some practices implemented policies and strategies to ensure all chemotherapy orders are signed and routed to the pharmacy in advance and, pending laboratory results and assessments, are within normal parameters. These strategies tended to come from those in management roles and aimed to alter physician behavior:
We have our docs . . . sign the orders 24 to 48 hours [in advance], and now they’ve started weekly rounds with the nurses, and they look at the next week’s schedule, and they say . . . “Don’t mix the chemo. I’ll sign it, but I want to see [the patients] the same day.”
Laboratory Testing Processes
Waiting for laboratory results before beginning infusions can delay the treatment start time, which is particularly salient if laboratory facilities are running behind schedule. Laboratory results are reviewed to indicate if the chemotherapy is safe to administer to the patient on the day of service. After the results are verified as within safe parameters, chemotherapy orders must be entered by the chemotherapy-privileged physician, signed, and sent to the pharmacy, which may also add to delay times, depending on the workload of physicians and pharmacists.
At one site, patients from rural areas often scheduled their chemotherapy infusions on the same day as their physician appointments and laboratory work to save travel time and distance. When the laboratory takes longer than usual to process results, a delay can occur:
[Patients] might think it’s hugely significant because they’re the ones sitting in the chair. Now we’ve drawn blood, and it’s been a lab issue where a line is down and two hours waiting for a count to result. And if you’re a five-hour treatment, that’s huge. (Infusion nurse)
We sometimes get labs within a half hour, and then the next time it will be over an hour. . . . Because when we’re sitting here, like today not very busy but yet we’re doing nothing, people look at us like, “Why aren’t you getting this done?” And it’s like it’s out of our hands. (Infusion nurse)
In response to laboratory processing times and chemotherapy treatments contingent on the patient’s results, practices are encouraging patients to have their laboratory work completed the day before their scheduled treatment (uncoupled visits) to prevent day-of-treatment delays. The uncoupling of laboratory work and infusion visits also facilitates efficient scheduling and chair assignment because any complications or holds in treatment can be addressed before the scheduled visit, and the chair schedule can be adjusted accordingly. Having the laboratory results to evaluate prior to the visit also ensures that up-to-date orders are prepared and approved to save time coordinating care on the day of treatment.
Despite the benefits of uncoupling visits, some patients experience difficulty coming into the practice two days in a row because of challenges stemming from transportation, caregiver schedules, and work schedules. Infusion center administrators face challenges trying to balance patient preferences with maintaining efficient operations and schedules at their practices. The situation is increasingly complex for patients traveling longer distances:
The physicians and providers or most everybody in the clinic are very sensitive to [patients having to come back] because these patients aren’t well, and they’re coming back and forth, and they’re traveling. That in and of itself is a delay that comes from a compassionate place. Yet it impacts our daily operations in a very huge way.
Discussion
This study examined factors that influence delays in chemotherapy delivery in ambulatory oncology practices. The authors found that delays in treatment were most associated with four specific themes: (a) discrepancies in care plans and missing orders for uncoupled visits, (b) undocumented and uncommunicated day-of-treatment order changes, (c) orders not signed in advance by physicians, and (d) laboratory testing processes. Ambulatory oncology practices that experienced the most frequent delays in patient care and treatment tended to experience challenges around clinician communication and coordination and organizational structures, such as unclear staffing roles and responsibilities and individual clinicians operating outside of standardized practice. The authors also identified strategies some practices used to prevent delays, such as particular clinicians being responsible for next-day order preparation, group huddles to coordinate upcoming patient treatments, practice policies to ensure physicians sign orders in advance of the patient’s visit, and uncoupling laboratory and treatment appointments.
These results align with previous studies that have found delays in chemotherapy delivery and wait times are caused at least in part by poor communication and coordination among clinicians in various areas of the practice, such as infusion, laboratory, and pharmacy (Bany Hamdan et al., 2018; Belter et al., 2012; Lamé et al., 2016; Lamm et al., 2015; Liang et al., 2015). As the findings suggest, plans of care are not always up to date on the day of infusion appointments. Infusion nurses do not rely solely on the electronic health record; they use other communication methods to clarify orders and prevent errors. This theme is consistent with an emerging topic of clinician communication research (Gross et al., 2016). Behavioral interventions that focus on improving teamwork, communication, and trust in ambulatory oncology settings have yielded positive outcomes. These strategies could be widely adopted and modified for individual practices (Bunnell et al., 2013).
The results of this study suggest that delays in chemotherapy delivery do not have a singular cause and do not occur in isolation; rather, they are compounded by the multilevel and multifocal organizational structure of ambulatory oncology practices. Other studies have shown similar findings. A study by Kallen et al. (2012) linked communication issues with unsigned orders. Delay times were decreased through a communication-based intervention that aimed to address unsigned chemotherapy orders through patient service coordinators who were responsible for contacting physicians with unsigned orders.
To ensure orders are prepared and a patient is ready for treatment, the authors found more practices adopting a model of uncoupled visits. Several studies have provided evidence that uncoupling laboratory draws and chemotherapy infusion appointments reduces delays (Dobish, 2003; Gjolaj et al., 2016). Gjolaj et al. (2016) created a new workflow for patients with chemotherapy infusion appointments to have their laboratory work drawn the day prior and orders signed that reduced infusion clinic wait times by about 22%. In addition, a study by Dobish (2003) found that adoption of next-day chemotherapy scheduling at outpatient clinics decreased wait times for patients and improved efficiencies for nursing and pharmacy.
However, as the study findings showed, patients’ access to laboratory services in rural areas and dependence on caregiver schedules may affect their preference for appointment uncoupling. The current findings also suggest that there is resistance from some physicians wishing to provide same-day service and see their patients before signing orders. One study suggested that methods to combat this resistance from physicians include communication, information sharing, and emphasizing patient safety (Dobish, 2003).
Limitations
In the original study, the survey phase did not include a quantitative measure of the day-of-treatment delays observed in the second, qualitative phase of the project. Such a measure would enable examination of the patterns, correlates, and consequences of delayed chemotherapy treatment. In addition, the absence of time and motion approaches hinders the ability to calculate empirically a prolonged appointment for chemotherapy. However, the study design provided an opportunity to observe care processes in situ to understand the complexities of chemotherapy treatment coordination and delivery. Although the authors included diverse oncology practices in the sampling frame, the findings may not be generalizable to all ambulatory oncology settings. The authors are also not able to infer causal relationships of concepts because of the cross-sectional study design. In the future, investigators could explore chemotherapy delays across a larger, more geographically diverse sample of practices. Examining the effects of delays on patient and caregiver outcomes, including time lost from work and time away from other responsibilities, may motivate additional quality improvement efforts to address this problem.
Implications for Nursing
Nurses are well suited to identify barriers to timely chemotherapy administration across diverse medical oncology settings because they are the clinicians primarily responsible for infusion services. Oncology nurses will benefit from structural and behavioral approaches to provide clarity surrounding oncology team members’ roles and functions that lead to timely chemotherapy delivery. Ambulatory oncology practices will benefit from workflows that allow all treatment plans to be finalized prior to the day of chemotherapy treatment, so nurses can focus on delivering timely, high-quality oncology care. 
Conclusion
The research presented in the current article uses a multimethod, qualitative approach to highlight and understand the causes and consequences of delays in chemotherapy administration. These findings suggest that clearly defined roles and functions within the ambulatory oncology team, as well as interventions to improve teamwork and communication in ambulatory oncology practices, will facilitate more timely chemotherapy infusion delivery. 
The authors gratefully acknowledge the patients and staff of the participating practices.
About the Author(s)
Megan Lafferty, PhD, is a qualitative methodologist in the Center to Improve Veteran Involvement in Care at the Portland VA Medical Center in Oregon; Alex Fauer, PhD, RN, OCN®, is a National Clinician Scholar at the University of California, Los Angeles; and Nathan Wright, MA, is a research associate and Milisa Manojlovich, PhD, RN, FAAN, is a professor, both in the School of Nursing, and Christopher R. Friese, PhD, RN, AOCN®, FAAN, is the Elizabeth Tone Hosmer Professor in the School of Nursing, a professor in the Department of Health Management and Policy, and director in the Center for Improving Patient and Population Health, all at the University of Michigan in Ann Arbor. This study was supported by the Agency for Healthcare Research and Quality Grant No. R01HS024914. Fauer was supported, in part, by a Doctoral Degree Scholarship in Cancer Nursing (133507-DSCN-19-048-01-SCN). The content is solely the responsibility of the authors and does not necessarily represent the official views of the U.S. Department of Health and Human Services. Lafferty, Fauer, Manojlovich, and Friese contributed to the conceptualization and design. Lafferty, Fauer, Wright, and Friese completed the data collection. Fauer provided the statistical support. All authors provided the analysis and contributed to the manuscript preparation. Fauer can be reached at ajfau@umich.edu, with copy to ONFEditor@ons.org. (Submitted November 2019. Accepted for publication January 21, 2020.)




