The Association Between Analgesic Treatment Beliefs and Electronically Monitored Adherence for Cancer Pain
Objectives: To determine whether clusters based on analgesic treatment beliefs among patients with cancer predict objective analgesic adherence.
Sample & Setting: 207 patients with cancer in the outpatient setting who were aged 18 years or older, self-identified as White or African American, were diagnosed with solid tumor or multiple myeloma, and were prescribed at least one around-the-clock analgesic prescription for reported cancer pain.
Methods & Variables: This study is a secondary analysis of an existing dataset. General linear modeling with a backward elimination approach was applied to determine whether previously identified analgesic treatment belief clusters, as well as sociodemographic, clinical, and pain variables, were associated with adherence behaviors.
Results: Significant explanatory factors were experiential in nature and included sociodemographic, clinical, and pain-related variables, explaining 21% of the variance in analgesic adherence. Analgesic belief clusters were not predictive of adherence.
Implications for Nursing: Future research should examine sociodemographic and other clinical factors, as well as the influence of analgesic treatment beliefs, to better understand adherence behaviors among patients with cancer.
Jump to a section
The majority of patients with cancer report pain, and as many as 38% of those patients report their pain as moderate to severe (Shi et al., 2011; van den Beuken-van Everdingen et al., 2016). Despite a lack of data on outcomes related to long-term opioid use for cancer pain (Meghani & Vapiwala, 2018), a number of cancer pain guidelines continue to identify opioids as a core component of moderate to severe cancer pain management (National Comprehensive Cancer Network [NCCN], 2020; Paice et al., 2016; World Health Organization [WHO], 2018). Although some pain management guidelines promote the use of complementary and alternative strategies (Dowell et al., 2016; NCCN, 2020), their affordability may be unmanageable for some, and several systematic reviews have noted insufficient evidence to support their clinical efficacy in alleviating cancer pain (Hetkamp et al., 2019; Kim, Kang, & Lee, 2018; Kim, Loring, & Kwekkeboom, 2018; Shin et al., 2016; Wayne et al., 2018). Therefore, analgesic use—and opioid use in particular—remains a primary modality for achieving moderate to severe pain control in the cancer population. Despite the many national initiatives that focus on advancing pain science from the provider perspective (Adams et al., 2017; Bonnie et al., 2017; National Academy of Medicine, 2020), little is known about the predictors of patients’ actual analgesic-taking behaviors.
As a result of the opioid epidemic and its healthcare implications, the phenomenon of analgesic adherence requires better understanding. A number of individual, family, provider, and system-level factors have been shown to predict nonadherence behaviors in this context (Rosa et al., 2020). Patients who experience less pain relief with analgesic use or higher side effect severity are typically less adherent to prescribed analgesic regimens, whereas those prescribed a strong opioid (classified as step 3 on the WHO analgesic step ladder) or an extended-release opioid show higher levels of adherence (Meghani et al., 2015). Hesitancy to use analgesics among family caregivers, as well as family characteristics, can also influence the analgesic adherence behaviors of patients with cancer pain (Lee et al., 2015; Schumacher et al., 2014b). Provider hesitancy to prescribe opioids and systemic/structural variables, such as socioeconomic status, race, and insurance coverage, have all been correlated with analgesic adherence to prescribed regimens (Meghani et al., 2013, 2014; Rhee et al., 2012; Valeberg et al., 2016; Wandner et al., 2014; Wieder et al., 2014).
Patient beliefs have also been shown to predict adherence behaviors. For example, patients who believed their doctor should focus on curing their illness over treating pain demonstrated lower analgesic adherence (Meghani et al., 2015). Inconsistent analgesic adherence for cancer pain has been correlated with increased hospitalization rates (Meghani & Knafl, 2016), as well as poor pain and quality-of-life outcomes (Manzano et al., 2014).
Understanding how patient beliefs inform decisions to adhere to prescribed analgesics is an important aspect of improving pain management strategies and subsequent outcomes (Miaskowski et al., 2001). Researchers have identified distinct patient clusters based on how patients make trade-offs (e.g., type of analgesic, type and severity of side effects, amount of expected pain relief, out-of-pocket costs) in considering analgesic treatment for cancer pain (Meghani & Knafl, 2017). Using choice-based conjoint analysis, Meghani and Knafl (2017) found that a majority of patients may be motivated predominantly by a single salient concern in their decision to use analgesics for cancer pain.
The current study builds on the data analysis of a previous study, which identified two unique clusters of patients based on how they prioritized their beliefs about analgesic treatment for cancer pain (Rosa et al., 2020). These analgesic treatment belief clusters were identified using a decision-making trade-off methodology, known as maximum difference (MaxDiff) scaling, to elicit which beliefs were most important to patients when considering analgesic use for the management of cancer pain. Patients in this study were most likely to trade off based on the belief that taking pain medicine would keep them from knowing what is going on in their body; the belief that patients who take analgesics for cancer pain become addicted was only moderately important across the sample (Rosa et al., 2020). The specific aim of the current study was to assess whether these unique analgesic belief clusters predicted objective analgesic adherence using an electronic medication monitoring system while accounting for relevant confounders.
Methods
Design and Study Population
This study is a secondary analysis of existing data from a parent study (RC1-NR011591, principal investigator: S.H. Meghani). The goal of the parent study was to explain racial and ethnic disparities in cancer pain outcomes, particularly to elicit trade-offs that patients with cancer pain employ while making cancer pain treatment decisions (using choice-based conjoint analysis) and their actual adherence to scheduled analgesic treatment using electronic monitoring with the Medication Event Monitoring System® (MEMS®) (Meghani et al., 2013, 2015). The parent study used a prospective observational design with repeated measures at baseline (T1) and three-month follow-up (T2). Patients were recruited from two outpatient medical oncology clinics at the University of Pennsylvania Health System in Philadelphia between December 2009 and August 2011. The institutional review board (IRB) of the University of Pennsylvania approved the parent study, and all participants provided written informed consent.
The current study was deemed exempt by the University of Pennsylvania IRB because all protected health information was removed from the dataset prior to study commencement. Consistent with the parent study, patients were eligible for the study if they were aged 18 years or older, self-identified as African American or White, reported a diagnosis of multiple myeloma or solid tumors, endorsed cancer pain, and had been prescribed at least one around-the-clock (ATC) oral analgesic. Patients using transdermal opioid delivery systems, such as fentanyl, were not included in the parent study sample because of MEMS vial limitations. The current study included 207 self-identified African American and White patients. A participant recruitment flow chart from the parent study was previously published, and a 14% attrition rate was noted between T1 (n = 241) and T2 (n = 207), with no statistically significant attrition identified by participants’ health status or race (Meghani et al., 2015).
Measures
Analgesic adherence: MEMS was used to electronically measure objective analgesic adherence. MEMS is a medication bottle cap that uses a microprocessor to record the event and time of a bottle opening in real time. Dose adherence, which was the primary measure of adherence to ATC analgesics, was defined as the percentage of the total number of prescribed doses that were taken by a patient. For instance, if a patient took 80 of 100 prescribed doses during the study period, dose adherence would be 80%. The procedures for calculating dose adherence were previously described in the parent study (Meghani et al., 2015). Investigators in the parent study performed sensitivity analysis to account for the observer effect (e.g., modified analgesic-taking behavior because of one’s awareness of being observed) and compared MEMS dose adherence from the total number of study days to MEMS dose adherence if the first 30 days of observation were removed (Meghani et al., 2015). Significant Spearman correlations for all patients in the sample (p < 0.001) suggested strong internal consistency between total dose adherence scores for the study duration and total dose adherence scores minus the first 30 days of observations. Based on these findings (Meghani et al., 2015), the MEMS dose adherence scores measuring all monitored days in the parent study were selected for use in the final analysis of the current study.
Index analgesic: ATC analgesics (index medications) were self-reported by patients during the T1 baseline interview and confirmed through a review of electronic health records. Index analgesics were classified per WHO’s (1986, 1996) analgesic step ladder. Categories include step 1 (nonopioids [e.g., nonsteroidal anti-inflammatories, such as ibuprofen or acetaminophen]), step 2 (weak opioids [e.g., codeine, tramadol]), and step 3 (strong opioids [e.g., methadone, oxycodone]).
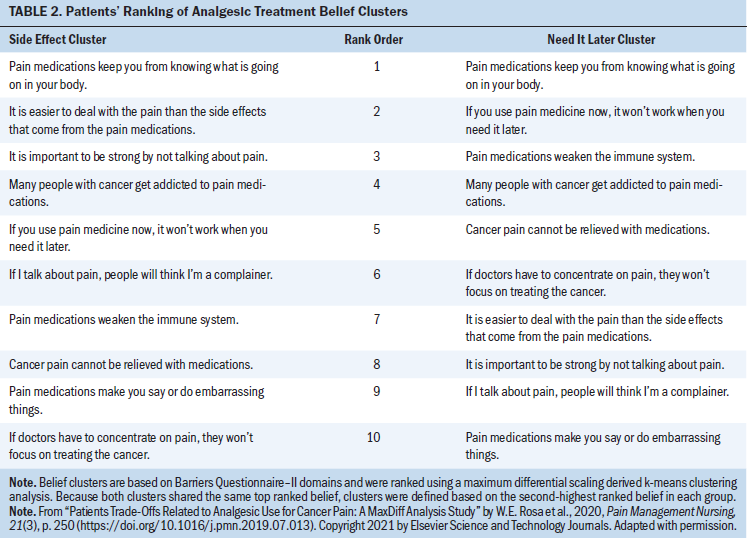
Analgesic beliefs for cancer pain: MaxDiff scaling was used to derive patient clusters based on beliefs about analgesic treatment for cancer pain. Belief clusters were originally identified using MaxDiff statistical techniques on JMP® Pro, version 14, software as described in a previous study (Rosa et al., 2020). MaxDiff is a trade-off methodology rooted in random utility theory (Thurstone, 1927). MaxDiff permits researchers to elicit increased choice discrimination through forced trade-offs between items and prevents scale use bias by requiring participants to make clear choices rather than merely rating preference strengths used in other ranking or discrete choice methods (Louviere et al., 2010, 2015; Marley & Flynn, 2015). MaxDiff data were then subjected to a k-means clustering analysis. Two unique clusters were identified: cluster 1 (n = 53) and cluster 2 (n = 154), which correlated with distinct analgesic treatment preferences. Because both clusters shared the same top-ranked analgesic belief, cluster 1 was named the “side effect” cluster and cluster 2 was named the “need it later” cluster based on the second-highest ranked preference of each group. Additional information on the rationale for the two-cluster model is provided in a previous publication (Rosa et al., 2020).
Self-reported barriers to analgesic use: The Barriers Questionnaire–II (BQ-II) (Ward et al., 1993) is a 27-item instrument that was used at T1 to assess patient beliefs and concerns about cancer pain management. The BQ-II is comprised of the following eight domains related to pain management concerns: (a) fear of addiction, (b) fear of tolerance, (c) fear of side effects, (d) fatalism about cancer pain, (e) desire to be a good patient, (f) fear of distracting healthcare providers from treating cancer, (g) fear of immune system impairment through analgesic use, and (h) concern about analgesic use masking patients’ ability to monitor the physiologic symptoms of their illness. The BQ-II has demonstrated strong internal consistency, with a Cronbach’s alpha of 0.89 (Ward et al., 1993) and 0.86 in the current study.
Analgesic side effects: The Medication Side-Effects Checklist (MSEC) (Ward et al., 1998) was used to capture side effects of analgesics at T1. The MSEC identifies the presence, type, and severity of eight analgesic side effects during the prior week (on a scale ranging from 0 [no severity] to 10 [extreme severity]). Side effects include constipation, drowsiness, nausea, vomiting, confusion, dry mouth, upset stomach, and itching. The MSEC has excellent internal consistency reliability, with a Cronbach’s alpha of 0.81 (Ward et al., 1998) and 0.79 in the current study.
Pain severity and pain impact: Baseline measurement of pain severity and impact were elicited at T1 using the Brief Pain Inventory (BPI) (Cleeland & Ryan, 1994). The BPI measures worst, least, and average pain scores during the week prior to assessment, as well as current pain level (on a scale ranging from 0 [no pain] to 10 [pain as bad as you can imagine]). The BPI has been well-documented in previous research of patients with cancer and has demonstrated internal consistency and reliability, with a Cronbach’s alpha ranging from 0.77 to 0.91 (Anderson et al., 2000; Cleeland et al., 1994; Cleeland & Ryan, 1994; Meghani et al., 2015; Meghani & Keane, 2007; Rhee et al., 2012; Yeager et al., 2019). In this study, the reliability coefficient was 0.9.
Pain Management Index: The Pain Management Index (PMI) was calculated for each patient according to WHO (1986, 1996, 2018) guidelines for cancer pain treatment. PMI is reflective of the relationship between the most potent analgesic prescribed and the patient’s self-reported pain level and is calculated by taking the most potent prescribed analgesic and subtracting the patient’s self-reported pain level (classified as mild, moderate, or severe). Insufficient analgesic prescription strength relative to a patient’s self-reported pain level is typically demonstrated by a negative PMI score.
Social support: A 6-item abbreviated version of the 27-item Social Support Questionnaire (Sarason et al., 1983) was used to identify patients’ levels of social support and satisfaction with their perceived support. Patients first identified the individuals in their life who provide social support and then rated the level of satisfaction level they experienced with the support. Information is elicited through questions such as, “Whom can you really count on to be dependable when you need help?” The rating scale ranges from 1 (very dissatisfied) to 6 (very satisfied), with higher scores reflecting more satisfaction with support.
Demographic and illness-related variables: Demographic data were self-reported and included age, gender, self-identified race, marital status, education level, income, and health insurance type. A number of variables related to illness were gathered through medical chart review, including cancer type and stage, time since cancer diagnosis, history of depression or substance abuse, and comorbidities, which were used to calculate a Charlson comorbidity score (Charlson et al., 1987).
Statistical Analyses
All analyses for the current study were performed using Stata®/IC, version 15. Descriptive statistics were generated for relevant sociodemographic and clinical variables. Means and standard deviations are provided for continuous variables and frequencies and percentages for categorical variables.
General linear modeling was the primary statistical method used to achieve the study aim. Prior to building the regression model, bivariate analyses between predictor variables and the outcome adherence variable were assessed. Relevant sociodemographic and clinical variables that were significant at the bivariate level (p < 0.2) at T1 were considered as potential predictors of MEMS dose adherence at T2. Two models were then constructed.
For model 1, variables that met the criteria, in addition to theoretically salient variables (e.g., history of substance abuse and presence of depression), were used to construct a preliminary prediction model employing a backward elimination method. The backward elimination method is useful in evaluating the value of each potential predictor when studying a phenomenon that may be influenced by a number of confounders (Cohen & Cohen, 1983). After starting with all individual potential predictors in the preliminary model, any variable that improved the model most significantly by its deletion was subsequently removed. This elimination process included theoretically salient variables. Theoretic variables are those that were identified as significant in explaining analgesic adherence behaviors in previous studies. This process was repeated until no additional model improvement was possible and all predictors were significant at the a = 0.05 level.
In model 2, the same theoretically salient variables used in model 1 were included. The clusters variable was then entered as a dichotomous categorical variable, with cluster 1 and cluster 2 as levels to evaluate the impact of analgesic treatment beliefs on analgesic adherence by observing any change in the R2 value. The clusters variable was the primary variable of interest and, therefore, was retained in the backward elimination process, regardless of statistical significance. This is consistent with the statistical convention to maintain insignificant findings in a final model when the explanatory variable is of primary interest or there is a specific hypothesis about a given variable (Grace-Martin, 2020; Heinze & Dunkler, 2017).
Variance inflation factors suggested low levels of multicollinearity among predictors in both models (1.49 and 1.43, respectively) (Chatterjee & Yilmaz, 1992). Using studentized residuals during residual analysis, no observations fell beyond the criteria of concern (x > 3, x < –3). The outcome variable was assessed using histograms and Shapiro–Wilk test (0.87), neither of which showed concerns with violations of normality assumptions. In addition, MEMS dose adherence was subjected to a sensitivity analysis to remove two observations significantly greater than 100% adherence, which did not change the Shapiro–Wilk value. 
Results
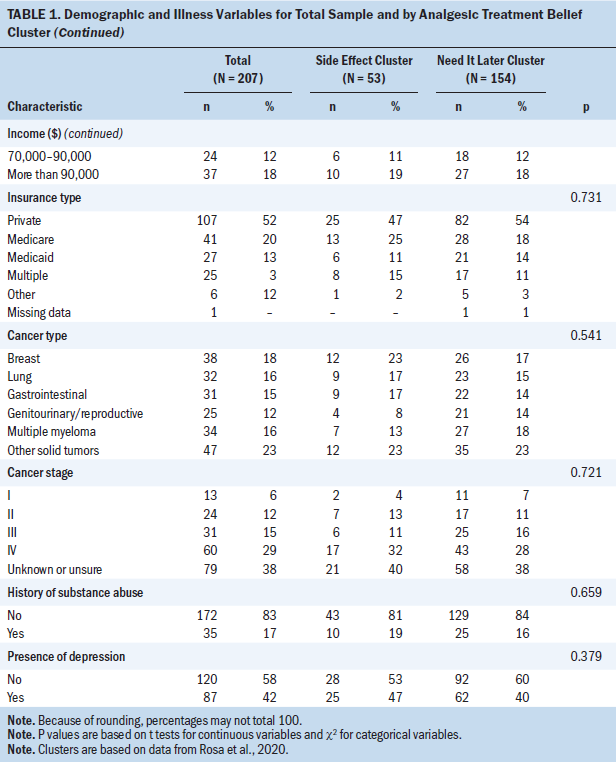
Patients (N = 207) had a mean age of 53.8 years (SD = 11.1), and most were female (n = 117). Most patients identified as White (n = 121), and less than half identified as African American (n = 86). The majority of patients rated their general health as good (n = 63) or fair (n = 77); only nine patients rated their general health as excellent at T1. Most patients denied a history of substance abuse (n = 172) or current presence of depression (n = 120). Table 1 shows demographic data and illness variables for the entire sample and by cluster. No significant differences between clusters were identified. Table 2 shows the belief clusters identified in the authors’ previous study and differences in their rank order (Rosa et al., 2020).
The analgesic and pain management variables among the sample (see Table 3) showed that patients used about 2.1 analgesics to treat their pain on average, with the vast majority of patients (80%) being prescribed a strong opioid (WHO step 3). Average pain scores during the prior week ranged from 3.4 to 6.9 (on a scale from 0 [no pain] to 10 [worst pain]). Clusters 1 and 2 differed significantly in how they rated severity of side effects (p = 0.043). There were no statistically relevant differences between clusters in terms of other variables, such as PMI, pain interference, or the number of self-identified barriers to analgesic use. 
MEMS Analgesic Adherence
The clusters variable based on analgesic beliefs was not found to be statistically significant at the bivariate level (p = 0.709) but was included in all modeling computation as the primary variable of interest. A number of relevant sociodemographic and clinical variables met inclusion criteria for linear modeling (p < 0.2) (see Table 4). Both average and worst pain scores were tested for potential inclusion; the average pain score (p = 0.004) was selected because of a higher level of significance. 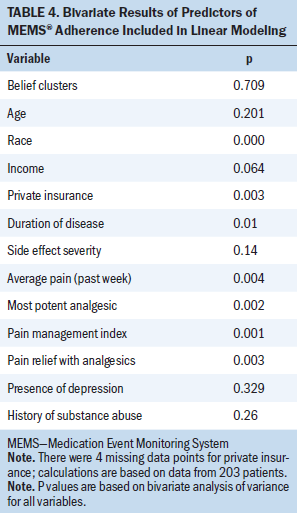
The first model showed that race, side effects, most potent analgesia prescribed, pain relief with analgesics, and the duration of disease were all significant at the a < 0.05 level. This analysis was initially generated without the cluster variable to evaluate the relationship between other salient correlates and the MEMS variable in the absence of cluster influence. Variables, such as average pain score, history of substance abuse, presence of depression, income, age, PMI, and insurance type, were excluded throughout the elimination process. About 21% of objective analgesic adherence variance using MEMS was explained by the final model (R2 = 0.207). The clusters variable was then entered to model 2 and, following the backward elimination method, the same variables were identified as statistically significant (see Table 5). Although the clusters variable was nonsignificant (p = 0.545), it remained in the model as the primary variable of interest. Similar to model 1, these predictors accounted for 21% of variance observed in the analgesic adherence variable (R2 = 0.208). 
Discussion
This study sought to determine whether previously identified unique clusters based on patients’ analgesic treatment beliefs (Rosa et al., 2020) predicted analgesic adherence behaviors that were objectively monitored electronically. Based on the results, analgesic belief clusters were not statistically associated with adherence in this adjusted analysis. However, other clinically relevant factors, such as race, side effects, most potent analgesia prescribed, pain relief with analgesics, and the duration of disease significantly predicted objective adherence to analgesics for cancer pain. The results raise important questions about the role of patient beliefs, sociodemographic background, and clinical history in relation to adherence behaviors in the setting of cancer pain.
Although analgesic treatment beliefs, preferences, and concerns of patients and families have been previously associated with analgesic use (Liang et al., 2013; Meghani et al., 2015; Meghani & Knafl, 2017; Rhee et al., 2012; Simone et al., 2012), the results of the current study show that beliefs ultimately do not explain patients’ objective analgesic adherence behaviors. For example, in an adjusted analysis accounting for other confounders, experiential variables (e.g., race, side effects, most potent analgesia prescribed, pain relief with analgesics, duration of disease) mattered the most to patients in predicting analgesic adherence. These experiential variables have all been substantiated by extant literature as having an impact on adherence behaviors (Manzano et al., 2014; Meghani et al., 2013, 2014, 2015; Meghani & Knafl, 2017). These covariates are clinically relevant and appear to be interrelated. For instance, stronger opioids may lead to improved pain relief but may also exacerbate the severity of side effects. In addition, stronger opioids may also relate to more advanced cancer diagnoses and increased adherence (Meghani et al., 2015; Oldenmenger et al., 2017).
Race was the most significant covariate within the model, which supports the findings of previous research (Meghani et al., 2013, 2014; Rhee et al., 2012). Studies exploring race related to analgesic adherence have demonstrated that African American and White patients differ on the beliefs and concerns most important to them. Previous analysis of the current study’s sample showed that African American patients were most concerned about severity of side effects, which has been positively correlated with increased nonadherence behaviors in this population (Meghani et al., 2013, 2015; Meghani & Knafl, 2017). White patients in the same sample tended to make trade-offs based on the amount of pain relief afforded by analgesic treatment. In previous findings, African American patients were less likely to be prescribed extended-release WHO step 3 opioids than White patients (Meghani & Knafl, 2017).
Severity of pain was not included in model 1 or 2 based on the analytic exclusion criteria but remains clinically significant. More potent analgesia may serve as a proxy for pain severity. In addition, disease duration likely serves as an indirect proxy for pain severity as it may imply more serious pain symptoms requiring higher potency analgesics. Of note, although theoretically salient, a history of substance abuse was also not a statistically significant variable in the final regression models. This may be because the majority of patients in the sample did not endorse a history of prior substance abuse.
Limitations
There are several limitations of this study. First, the analgesic belief clusters used as the primary variable of interest are not exhaustive of all potential patient beliefs. However, a well-validated tool was used to elicit pain management and analgesic concerns (Ward et al., 1993). Future research may aim to elicit additional relevant cancer pain treatment beliefs not included in the parent study instrument because they relate to analgesic use or new concerns garnering salience in the current national context (e.g., opioid prescribing, insurance coverage changes, focus on use of immediate-release opioids). Second, although clusters based on analgesic beliefs were not statistically significant, the results support further exploration of the extent to which patients’ beliefs interact with other clinical and sociodemographic variables to influence cancer pain outcomes. Third, although a two-cluster model was previously identified (Rosa et al., 2020), there are likely multiple groups of patients that prioritize their beliefs differently. The authors anticipate that the results of the current study are merely a starting point for identifying how variant patient priorities inform analgesic adherence in patient populations. Fourth, although the age of the data is a concern, the parent study researchers collected data at the peak of the first wave of the opioid epidemic and as heroin-related overdose deaths had started to spike during the second wave (Centers for Disease Control and Prevention, 2020). Therefore, the current authors expect that this unique longitudinal dataset focusing on patients’ analgesic usage has relevance in the current and evolving context of the U.S. opioid epidemic. Last, although these findings support previous research findings about predictors of adherence, the results cannot be generalized beyond this sample.
Implications for Nursing
Clinical nurses should observe for changing trends in opioid prescribing practices because they relate to analgesic adherence behaviors. Although certain guidelines specific for cancer pain management continue to recommend opioids based on patients’ subjective reports of pain and a combination of short- and extended-release opioids for optimal pain control (NCCN, 2020; WHO, 2018), the broader national conversation on opioid prescribing has been turning toward more modest analgesic treatment focused on immediate-release opioid use (Dowell et al., 2016). The results of the current study, in conjunction with the national opioid epidemic discourse, suggest that re-evaluation of interventions geared toward improving adherence for cancer pain is needed. Based on these results and the results of previous studies (Meghani & Bruner, 2013; Meghani et al., 2014; Oldenmenger et al., 2017; Rhee et al., 2012), there are subsets of patients that continue to require ATC analgesic prescriptions to effectively manage pain.
Of concern, previous data highlight that patients are not using analgesics for cancer pain on a scheduled basis. For example, previous findings related to this current sample found that only 69% of patients (n = 207) were adherent to WHO step 3 immediate-release opioids, and roughly 74% were adherent to extended-release opioids (Meghani & Knafl, 2017). Although there have been substantial efforts to tailor education for patients to address analgesic beliefs and barriers, systematic reviews show that these interventions improve neither analgesic adherence nor associated outcomes for cancer pain (Bennett et al., 2009; Oldenmenger et al., 2009, 2018). The evidence suggests that continued focus on evaluating key clinical variables, such as analgesic side effects and pain reporting, as well as other sociodemographic and economic factors like race, income, and health literacy, may be central to improved outcomes associated with cancer pain (Meghani & Bruner, 2013; Meghani et al., 2013, 2014, 2015; Meghani & Knafl, 2017).
Implications for Research and Policy
This study has numerous research and policy implications. First, additional qualitative research that provides more subjective data on analgesic beliefs and preferences during the opioid epidemic is warranted. Although there are many qualitative studies that have evaluated the phenomenon of analgesic adherence for cancer pain (Manzano et al., 2014; Schumacher et al., 2014a, 2014b), they largely rely on semistructured interviews. One study used a free-listing method and interviews to investigate opioid self-management practices for patients with cancer-related pain (Meghani et al., 2020). Methodologies, such as concept mapping and ethnographic observation (to the extent possible), may provide additional insights into the behaviors and contextual dynamics surrounding prescribed analgesic adherence behaviors. In particular, approaches that elicit analgesic beliefs and preferences among different groups of patients, families, and prescribers based on the experiences of cancer pain and analgesic treatment options may be helpful in understanding divergent priorities among other populations. Further qualitative inquiry may also shed more light on patient concerns not measured in this study and provide additional information on how beliefs interact with systemic/structural and family dynamics to influence analgesic adherence behaviors and cancer pain outcomes, which are multifactorial interactions discussed only minimally in previous studies (Schumacher et al., 2014a, 2014b).
Second, future research that explores complementary and integrative health approaches to cancer pain management is needed (Bao et al., 2014). Such interventions are supported by pain management guidelines (Dowell et al., 2016; NCCN, 2020); however, there is insufficient evidence to support their clinical efficacy in managing various types of cancer pain (Hetkamp et al., 2019; Kim, Kang, & Lee, 2018; Kim, Loring, & Kwekkeboom, 2018; Shin et al., 2016; Wayne et al., 2018). As pain outcomes data related to complementary and integrative health services are obtained through expanded research initiatives, policies that increase access to and plan coverage and affordability of these services—in conjunction with more traditional analgesic and intervention pain relief options—will be imperative to ensure pain care equity across patient populations (Office of the Assistant Secretary for Health, 2019).
Finally, continued efforts to clarify the association between patients’ analgesic beliefs and analgesic adherence behaviors are needed to further personalize care and improve safety and health outcomes. Although the two analgesic treatment belief clusters in this study were not statistically associated with objective adherence behaviors, there are likely multiple groups of patients that prioritize beliefs in different and nuanced ways, which informs their analgesic use accordingly. Future scientific inquiry should assess for these additional decision-making patterns. Policies that ensure timely pain management for any patient experiencing pain secondary to cancer are needed across institutional and system-wide settings. 
Conclusion
This study shows that patient clusters based on analgesic treatment beliefs do not significantly influence adherence behaviors. However, clinical variables were identified that speak to the experience of cancer pain and which pain treatments are most relevant to analgesic adherence. The results of this study affirm extant literature and support ongoing evaluation to address the key experiential variables that influence pain outcomes for patients with cancer through clinical, research, and policy interventions.
About the Author(s)
William E. Rosa, PhD, MBE, ACHPN®, FAANP, FAAN, is a postdoctoral research fellow in psycho-oncology in the Department of Psychiatry and Behavioral Sciences at Memorial Sloan Kettering Cancer Center in New York, NY; Barbara Riegel, PhD, RN, FAHA, FAAN, is the Edith Clemmer Steinbright Professor of Gerontology, Connie M. Ulrich, PhD, RN, FAAN, is the Lillian S. Brunner Chair and a professor of bioethics and nursing, and Jesse Chittams, MS, is a biostatistician and managing director and Ryan Quinn, MPH, is a biostatistician, both in the BECCA Lab in the Office of Nursing Research, all in the School of Nursing at the University of Pennsylvania in Philadelphia; and Salimah H. Meghani, PhD, MBE, RN, FAAN, is a professor and Term Chair of Palliative Care in the Department of Biobehavioral Health Sciences and NewCourtland Center for Transitions and Health and a senior fellow in the Leonard Davis Institute of Health Economics at the University of Pennsylvania. The data used in this study are from a National Institutes of Health/National Institute of Nursing Research Challenge Grant (RC1-NR011591) awarded to Meghani. During this writing, Rosa was supported by the American Cancer Society Doctoral Degree Scholarship in Cancer Nursing Grant (DSCN-19-054-01-SCN) and the Robert Wood Johnson Foundation Future of Nursing Scholars Program. Rosa is also supported by a National Institutes of Health/National Cancer Institute (NCI) Support Grant (P30 CA008748; NCI award number: T32 CA009461). Rosa, Riegel, Ulrich, and Meghani contributed to the conceptualization and design. Meghani completed the data collection. Chittams and Quinn provided statistical support. Rosa, Chittams, Quinn, and Meghani provided the analysis. Rosa, Riegel, Chittams, Quinn, and Meghani contributed to the manuscript preparation. Rosa can be reached at rosaw@mskcc.org, with copy to ONFEditor@ons.org. (Submitted March 2020. Accepted June 26, 2020.)




